1. Introduction
Renewable energy has attracted considerable research attention because of environmental pollution and the exhaustion of fossil fuels. Thermoelectric power generation (TEG) is a promising technology for sustainable energy because more than 60% of the energy is consumed as waste heat during energy generation and consumption. Moreover, TEG can be used to generate electricity from irregular heat sources over wide ranges of temperature.1) The TEG system is also advantageous in that it has no moving parts, is environmentally friendly, and can be integrated into other systems. However, there has been no commercialization of large-scale waste heat systems; moreover, the efficiency of TEG modules is limited because of the low energy conversion efficiency ZT (= σS2T/κ, where σ, S, T, and κ are the electrical conductivity, Seebeck coefficient, absolute temperature, and total thermal conductivity, respectively) of thermoelectric (TE) materials. Therefore, intensive research on TE materials has been conducted to improve their TE properties. Additionally, their mechanical properties have been studied to overcome harsh conditions such as thermal and mechanical stresses, thermal shock, and vibrations in automotive TE generators (ATEGs).
TE materials such as PbTe-, skutterudite-, half-Heusler-, silicide-, and SnSe-based alloys are the most important components determining the efficiency of TEG systems.2-6) Silicide-based TE materials (n-type magnesium silicide (Mg2Si) and p-type higher manganese silicide (HMS)) have received significant attention as ATEGs owing to their low density, low cost, and environmentally friendly constituent elements despite their relatively low TE properties.7) Unfortunately, the silicide-based TE materials have disadvantages such as low TE properties of HMS (ZT 0.5-0.6) and low fracture toughness (KIc) of Mg2Si (~ 0.82 MPa m1/2).8-11) These disadvantages must be overcome to expand application of these TE materials. Therefore, we need to investigate the TE and mechanical properties of silicide-based TE materials to simultaneously obtain a high conversion efficiency and mechanical reliability of the TE module.
There is a serious trade-off relationship between the TE and mechanical properties in silicide-based TE materials.12) Typically, nanocomposites and nanostructuring for TE materials effectively improve the TE properties by manipulating the electronic and thermal transport properties. However, silicide-based TE materials have an unfavorable trade-off relationship between σ and S or between σ and κ.13) Silicide-based TE materials have a narrow optimized carrier concentration (nc) range.14) The similar mean free path of the carriers and phonons limit the enhancement of TE properties.15,16) Therefore, the nanocomposites would be effective in improving the TE properties of the silicide-based TE materials via band engineering. Additionally, KIc is considerably influenced by the microstructure of a material and can be increased by hindering crack propagation via the introduction of nanophases. Consequently, nanocomposite strategies are more favorable than nanostructuring for improving the trade-off relationship of silicide-based TE materials.17)
To improve the trade-off relationship between the TE and mechanical properties, studies are required on the intrinsic properties of the matrix and nanophases, dispersibility, uniformity, morphology, and amount of the nanophase, and interfacial properties. In addition, it is necessary to optimize the fabrication process used to prepare the nanocomposites. Therefore, herein, we investigate the fabrication processes of nanocomposites to improve the trade-off relationship between the TE and mechanical properties of silicide-based TE materials.
2. Processing Technologies for Silicide Powders
In nanocomposites, the intrinsic properties of the matrix are important for determining the TE and mechanical properties. It is essential to establish appropriate synthesis methods and conditions.
Figure 1 illustrates the fabrication process of silicide-based TE powders. There are several fabrication processes, such as solid-state reaction, melting, arc melting, and melt spinning. The solid-state reaction (Fig. 1(a)) is the most conventional process for the preparation of polycrystalline solids. This process is dependent on various conditions such as temperature, time, surface area of the solid, reactivity, and thermodynamic free energy. Mg2Si-based compounds have been prepared by solid-state reactions owing to the large difference in the melting points of Mg and Si. Kim et al. prepared Mg2Si polycrystalline bulks co-doped with Al and Bi via a solid-state reaction and spark plasma sintering (SPS), and obtained a ZT of ~ 1.0 at 873 K.18) Based on these results, an up-scaled solid-state reaction process was achieved, which gave a ZT of ~ 0.98 at 873 K.19) Moreover, Liu et al. and Khan et al. synthesized Mg2(Si, Sn) and Mg2(Si, Sn, Ge) solid solutions with ZT > 1.0 via a solid-state reaction.5,20) This process is also suitable for HMS. Al-doped and Ge-doped HMSs were prepared via a solid-state reaction, and ZT values of ~ 0.5 and ~ 0.6, respectively, were obtained.9,21) However, as achieving a high-quality powder by this process requires the use of many experimental parameters, a sintering process is indispensable to obtain a single phase, indicating that it is disadvantageous in terms of performance reproducibility.19)
Melting (Fig. 1(b)) is widely performed for the fabrication of Bi2Te3-, PbTe-, and SnSe-based TE material ingots owing to their high homogeneities.2,22,23) However, it is not suitable for the preparation of silicide-based TE materials owing to the high melting points of Si and Mn and the vaporization of Mg.
Arc melting (Fig. 1(c)) is the widely used method for fabricating alloys. The electric arc between a tungsten electrode and raw materials in a copper hearth causes heating. Arc melting can easily increase the temperature to 2273 K, thereby shortening the process time. In general, melting is performed five times to increase the homogeneity of the melted ingots. Kim et al. performed arc melting to fabricate an HMS (Mn11Si19) ingot and found that this process can be used to control the secondary phase in the HMS matrix.24) Sadia et al. also investigated the texture anisotropy of HMS synthesized by arc melting and hot pressing (HP).25)
Melt spinning (Fig. 1(d)) is a rapid solidification technique. The alloy melted by an induction coil is dripped onto a wheel and cooled. This results in an unexpected microstructure, with nanoinclusions, secondary phases, and heterostructures in the melt-spun ribbon, which produce an enhanced power factor and reduced κlat. In silicide-based TE materials, nanostructured HMS was synthesized by melt spinning. Significant enhancement in the TE properties was achieved owing to the enhanced power factor and reduced κlat.26) However, Mg2Si-based TE materials present limitations when synthesized by melting-based processes (melting, arc melting, and melt spinning) owing to the vaporization of Mg. Therefore, it is necessary to precisely control the synthesis conditions in order to obtain high-quality powders.
3. Processing Technologies for Nanocomposites
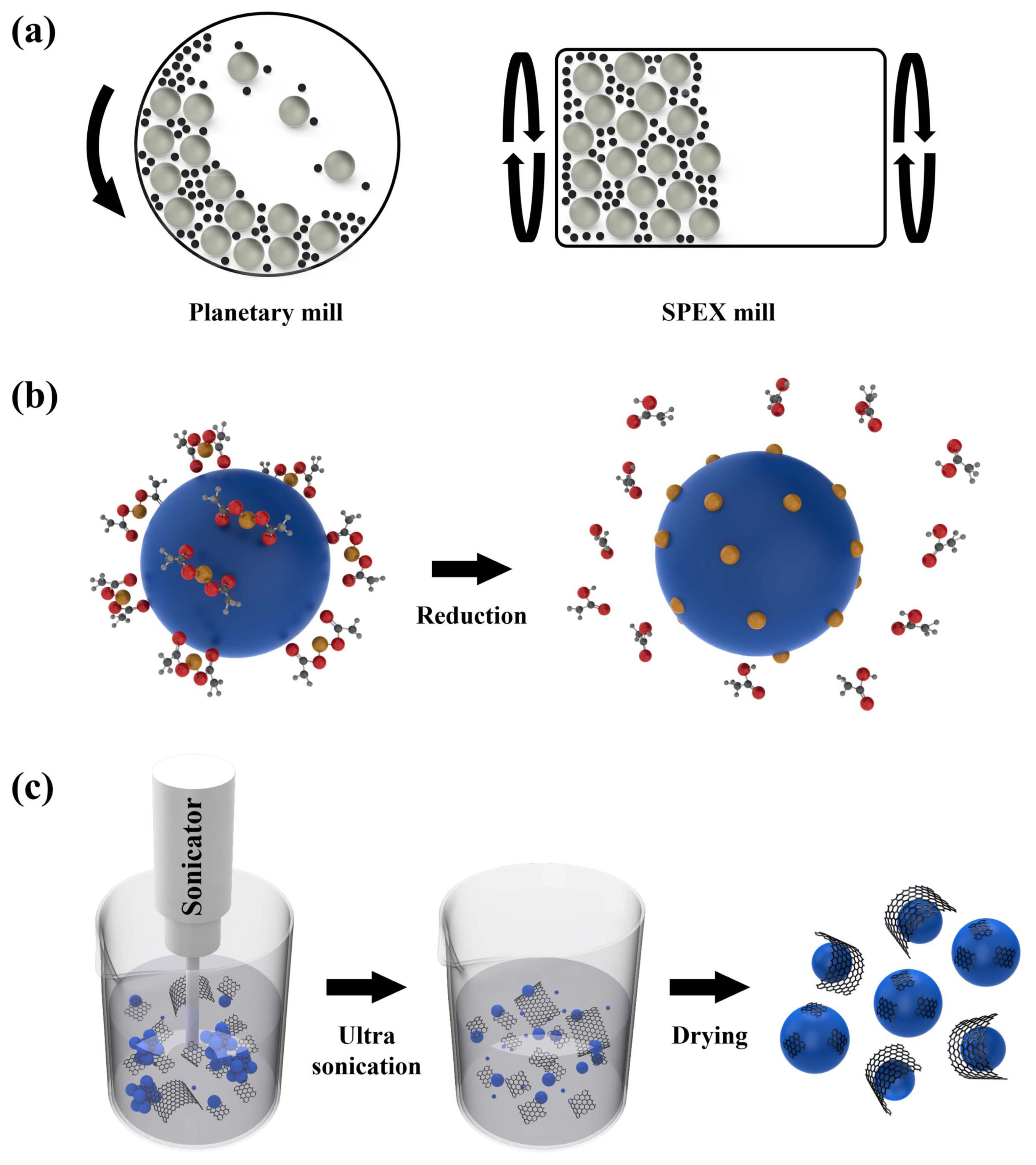
To obtain high TE and mechanical properties, we need to establish design rules for the fabrication of nanocomposites. Fig. 2 illustrates the typical nanocomposite fabrication processes. Ball milling (Fig. 2(a)) is a conventional process for grinding and blending materials. High-energy ball milling is a particularly effective method for fabricating hybrid powders and nanostructured powders. Poudel et al. realized a nanostructured p-type BiSbTe alloy with high TE properties via ball milling.27) However, this process is mainly used to homogeneously mix each material (matrix and nanophases) in silicide-based TE nanocomposites. Many researchers have employed this process to uniformly introduce nanophases (SiC, carbon nanotubes (CNTs), CaO, Si, and TiO2) in the Mg2Si matrix.28-32) Moreover, we fabricated heterostructured nanocomposites of HMS and metal silicides using this process.
The introduction of nanoparticles is a promising strategy for improving the TE and mechanical properties of silicide-based TE materials. A mix-and-heat process (Fig. 2(b)) can be used to easily decorate metal nanoparticles onto the surface of powders; for this, a metal acetate without any solvent and electric current are required. Ball milling is required to attach the metal acetate to the surface of the powder, and heat treatment is required to reduce the acetate (CH3COO−). Variables, such as the type and loading amount of the metal acetate, reduction heat-treatment temperature, time, and heating rate, determine the size and dispersibility of the metal nanoparticles.33) However, this process is disadvantageous in that it is difficult to completely control the uniformity of the metal nanoparticles. Using this process, metal nanoparticles several tens of nanometers in size were introduced into the silicide-based TE powders, which affected the TE and mechanical properties.11,12,34)
Wet chemical mixing is a method for blending powders and nanophases uniformly. As this process is carried out in a solvent, it can effectively increase the uniformity of the nanophases and prevent agglomeration of the matrix and nanophases, compared to other processes. In this process, solvents that do not react with the matrix and nanophases should be identified, and proper drying conditions should be established. Using this process, microstructures of reduced graphene oxide (rGO), CNT, and SiC-wrapped or -decorated powders were obtained, which confirmed the change in the TE and mechanical properties.14,29,35)
In nanocomposites, it is important to determine the optimal conditions for fabrication because the dispersibility, uniformity, size, shape, and amount of the nanophases, as well as the intrinsic characteristics of the matrix and nanophases, are important design rules for determining the TE and mechanical properties.
4. Processing Technologies for Sintering
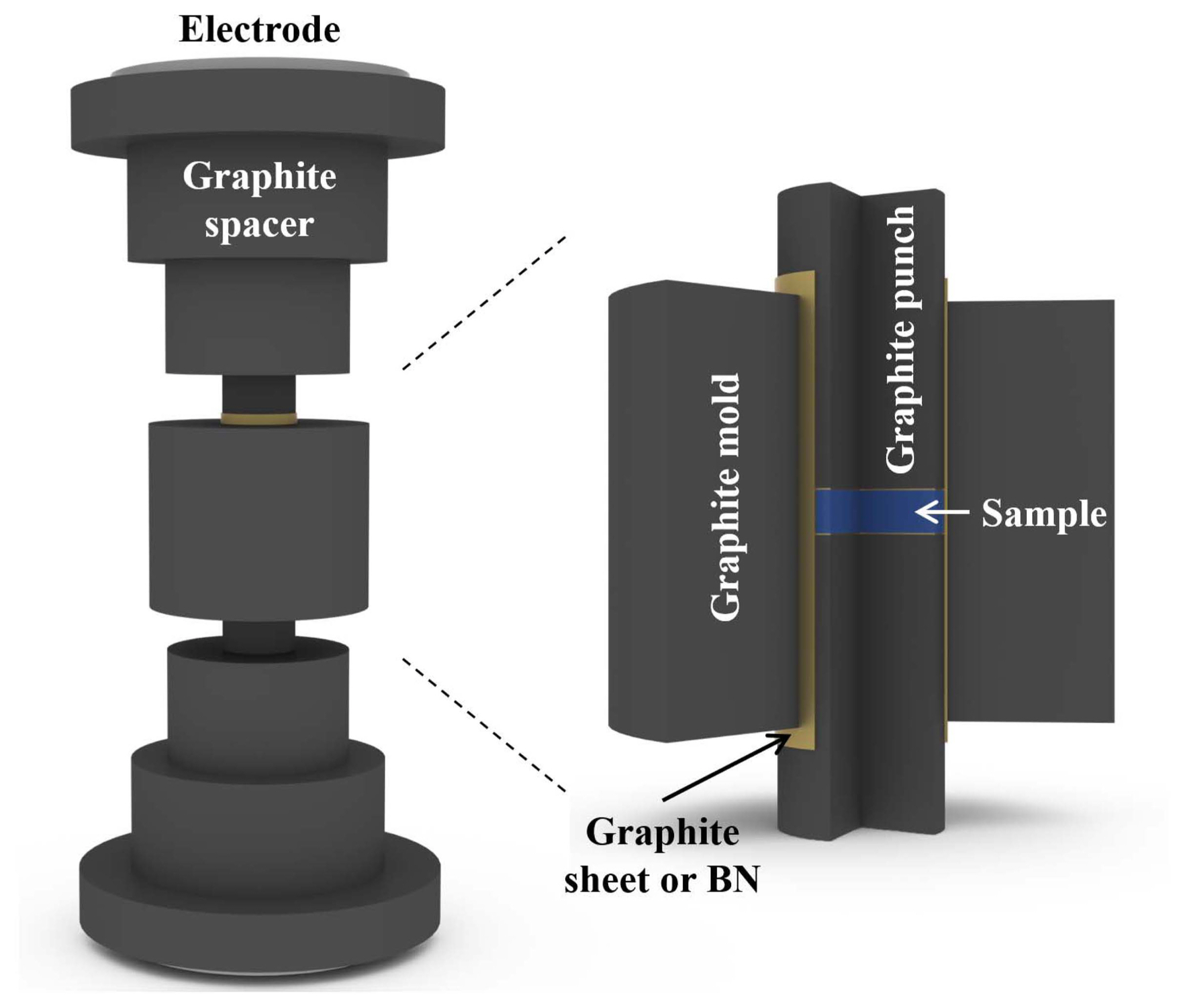
SPS and HP are typical sintering methods for TE materials. SPS is a novel technique using a pulsed direct electric current passed through the sample in a graphite mold. Joule heating in the graphite mold plays a dominant role in the densification of the powder. It employs very high heating and cooling rates (100 K/min). In contrast, HP employs low heating and cooling rates because the heat is provided by an external heating source. Therefore, SPS is advantageous for densifying TE powders with a nanostructure.
Precise control of the sintering conditions is required to obtain high-density compacted polycrystalline bulk TE nanocomposites. Variables such as sintering temperature, time, and pressure need to be considered to maintain the nanostructure in the matrix. For powder densification, the sample needs to be prepared in a graphite mold (Fig. 3). For silicide-based TE materials, in particular, as the sample is easily broken by thermal shock at a high heating rate, it is necessary to insert a carbon sheet into the material for thermal shock relaxation. Many researchers have employed SPS to obtain compacted silicide-based TE nanocomposites.14,34,35) However, silicide-based TE nanocomposites with nanophases such as CNT and SiC, which have high stability at high temperatures, may require HP to increase their sintered densities.28,29)
5. Microstructures of Various Nanocomposites
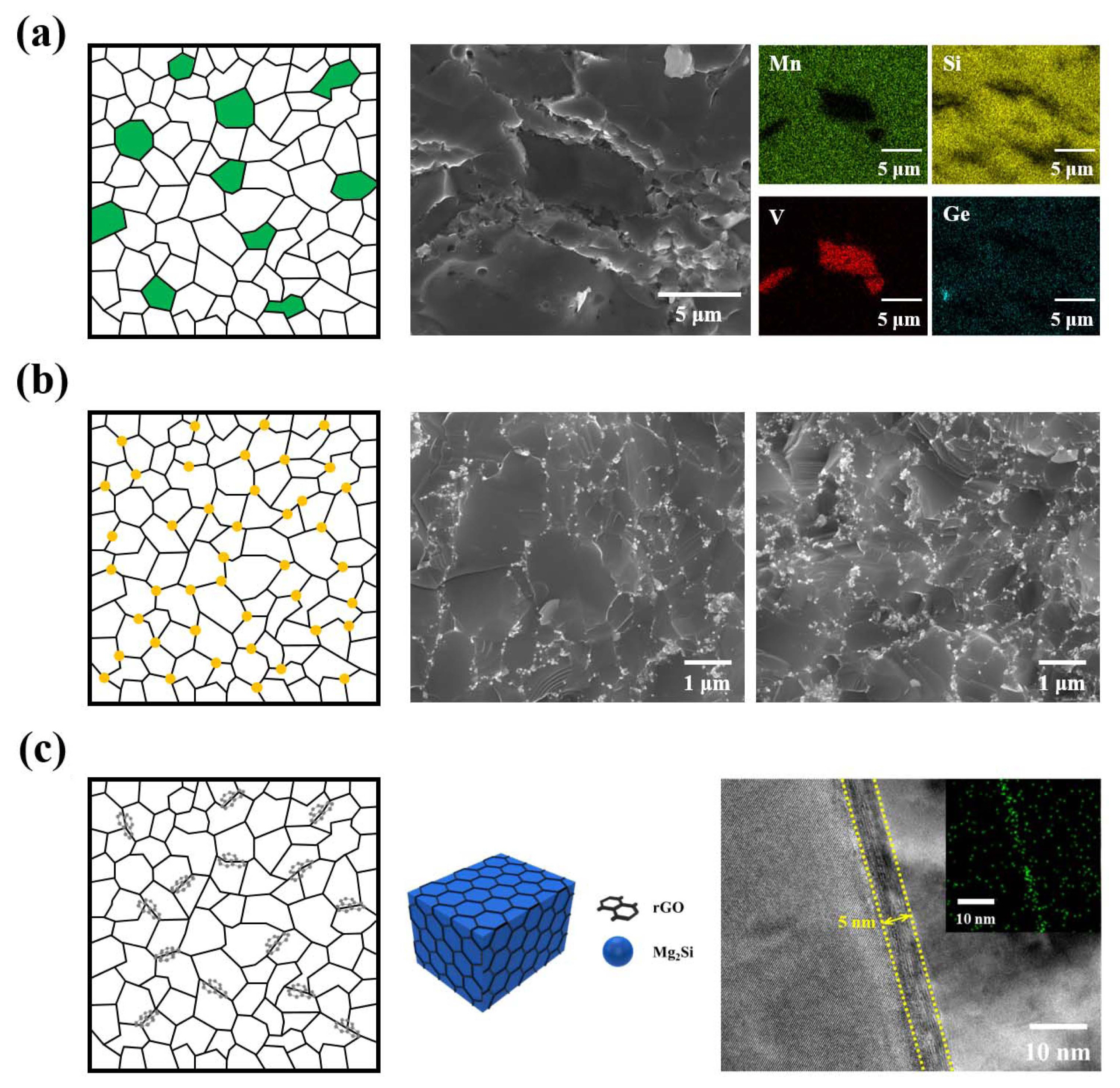
A microstructure design rule for nanocomposites is a prerequisite to obtaining high TE and mechanical properties. In particular, silicide-based TE materials require precise control of the band structure and microstructure because of the limitation in the enhancement of TE properties via nanocomposite formation. The heterostructure (Fig. 4(a)) plays an important role in enhancing the TE properties by increasing the power factor and reducing κlat. Control of the interfacial properties is also essential to manipulating the electronic and thermal transport properties. We have studied the heterostructure between HMS and metal silicide and have significantly increased the power factor of the composites by enhancing S via interfacial band engineering. Moreover, HMS composites incorporating MnS improved the TE properties through compositional tuning, S doping at Si-sites, and the heterostructure between HMS and MnS.36) However, they are not effective in improving the KIc because the micro-scale composites do not effectively prevent crack propagation. To improve KIc, it is necessary to introduce a nanophase with high mechanical strength. Figs. 4(b) and 4(c) show a microstructure in which nanoparticles or nanowires and nanosheets are incorporated. The uniformly distributed nanophase can interfere with crack propagation, and for nanowires and nanosheets, can absorb the crack propagation energy and prevent crack propagation in the matrix more effectively. An increase in KIc was confirmed by introducing metal nanoparticles, rGO, and SiC nanowires; low-dimensional nanophases such as rGO and SiC nanowires were more effective in improving KIc owing to their high interface densities.14,35) However, for Mg2Si, the TE properties were maintained or reduced when a nanophase was introduced owing to the influence of the intrinsic properties of the nanophase and the similar mean free paths of electrons and phonons.15,17) Additionally, the low work function of Mg2Si (~3.59 eV) limits the availability of suitable nanophases for band alignment matching, indicating that reduction in μHall is inevitable.37) On the other hand, HMS has a relatively moderate work function (~ 4.78 eV); therefore, an increase in the power factor and decrease in κlat could be expected on introduction of nanophases. We have been studying HMS with Fe, Co, and Ni nanoparticles. We obtained an increased power factor due to carrier transfer and energy filtering caused by proper band alignment between the metal nanoparticles and HMS. Additionally, Li et al. greatly increased the TE properties by minimizing the reduction in the power factor and reducing κlat to the theoretical limit by introducing an MnTe nanophase into HMS.38)
The enhancement of the TE and mechanical properties of nanocomposites is strongly dependent on their microstructure and band structure, and it is essential to establish optimal conditions to obtain a high performance. Therefore, it is necessary to continuously study the TE properties and mechanical reliability of silicide-based TE nanocomposites to fabricate high-efficiency TE modules.
6. Summary
This review summarizes the preparation and design rules of silicide-based thermoelectric bulk nanocomposites with a high trade-off relationship between the thermoelectric properties and mechanical reliability. The nanocomposites could potentially enhance the thermoelectric performance of silicide-based thermoelectric materials via manipulation of electronic and thermal transport properties. Moreover, they can effectively improve the fracture toughness by inhibiting crack propagation. A novel microstructure for the enhancement of thermoelectric and mechanical properties of silicide-based thermoelectric materials was investigated experimentally using various fabrication processes. However, establishing effective design rules for silicide-based thermoelectric nanocomposites with high ZT and mechanical reliability remains a challenge. We believe that our perspectives on the development of novel thermoelectric nanocomposites will significantly benefit automotive thermoelectric generator applications.