Ko, Kang, Cheong, and Yoon: Recent Progress in Cathode Materials for Thermal Batteries
Abstract
Thermal batteries are reserve batteries with molten salts as an electrolyte, which activates at high temperature. Due to their excellent reliability, long shelf life, and mechanical robustness, thermal batteries are used in military applications. A high-performance cathode for thermal batteries should be considered in terms of its high capacity, high voltage, and high thermal stability. Research progress on cathode materials from the recent decade is reviewed in this article. The major directions of research were surface modification, compounding of existing materials, fabrication of thin film cathode, and development of new materials. In order to develop a high-performance cathode, a proper combination of these research directions is required while considering mass production and cost.
Key words: Thermal batteries, Cathode materials, High capacity, High voltage, High thermal stability
1. Introduction
Thermal batteries are reserve batteries that can be stored for a long period in an inactive state, which can then be activated to generate power when needed. Thermal batteries include molten salts as an electrolyte and employ an internal pyrotechnic source to bring the battery stack to the operating temperature. Thermal batteries are high-temperature power sources that typically operate between 350 and 550°C. Thermal batteries are used for many military applications, such as power sources for guided missiles and proximity fuses in ordnance devices because of their excellent mechanical robustness, reliability, and long shelf life 1). The progress of research on thermal batteries until about 2006 is summarized in the review by Masset et al.. 1-5) This review provides an overview of molten salt electrolytes, 2) cathode materials, 3,4) and molten salt electrolytes 5) of thermally activated batteries. As a representative thermal battery electrode, Li-Si is used as an anode and FeS 2 (pyrite) is used as a cathode; LiCl-KCl is used as an electrolyte. Fig. 1 shows the basic structure of an Li-Si/LiCl-KCl/FeS 2 thermal battery. So far, various studies have focused on optimizing each component in these systems. 6-9)
Among them, studies focusing on the cathode are largely divided into materials and manufacturing methods. The most commonly used cathode material to date is FeS 2. In addition to studies on optimizing electrode fabrication using FeS 2, 10-15) studies have been conducted on the use of materials such as CoS 2, 16-21) NiS 2, 22-24) NiCl 2, 25-28) CuVO, 29-30) and others. 31-33) Masset et al. cited the following key properties to be considered as cathode materials for thermal batteries: 4)
Redox potential: it should have a discharge potential that is compatible with the electrochemical window of the electrolyte in order to prevent oxidation. Ability to provide a fixed discharge plateau: it should undergo multiphase discharge and not intercalation. High thermal stability: thermal decomposition and associated possible chemical reactions caused by decomposition products should be minimized (e.g., S2 in the case of FeS2 reacting with the anode or pyrotechnic source in the battery). Electronically conductive: the resistance of the cathode should be minimized. Low solubility of the cathode materials in molten electrolytes: self-discharge reactions with attendant loss in capacity should be prevented. Low solubility of discharge products in the molten electrolytes: prevent self-discharge reactions. Stable towards moisture and/or oxygen: to prevent oxide production at the cathode surface. Ability to be wetted by electrolyte: this minimizes the contact resistance at the electrolyte (separator)/electrode interface. Good discharge kinetics (high exchange-current density): provides high rate capability. Reasonable costs. Being environmentally friendly is an additional desirable attribute.
The second area of focus in prior research is on the method for manufacturing the cathode electrode. Generally, the constituent materials are produced in the form of pellets with a thickness of several hundreds of micrometers using a powder compaction process. However, the formed pellets are thicker than the optimum thickness in terms of utilization of the electrode material because they must maintain a minimum mechanical strength. For these reasons, an electrolyte material is added to the electrode to compensate for the lower utilization of the electrode material. However, this approach makes the electrode becomes thicker and reduces the ratio of the active material in the electrode. When the thickness of the electrode pellet is greater than a certain level, electrode utilization decreases significantly, which may decrease the energy density of the thermal battery. To solve such a problem, it is essential to choose a process for thinning the electrode such that the electrode has sufficient mechanical strength with optimal electrochemical performance. Therefore, some studies focused on the use of a tape casting process to thin the cathode of a thermal battery. 11-13)
In this review, the cathode materials of thermal batteries are reviewed in terms of material types and electrode manufacturing methods. The scope of this review is the past 10 years since the reviews published by Masset et al.. 1-5)
2. FeS2
The intrinsic difficulties of engineering a thermal battery based on Ca/CaCrO 4 electrochemistry were obviated with the introduction of Li-alloy/FeS 2 couples. 1) The performance of the Ca/CaCrO 4 electrochemical system tended to be somewhat unpredictable. 1) FeS 2 was readily obtained from processing pyrite, which is a plentiful, inexpensive material compared to chemically synthesized CaCrO 4. In addition, FeS 2 has higher electrical conductivity at elevated temperatures. This provides improved power and lifetimes over the Ca/CaCrO 4 system. 3,4) For these reasons, the Li-alloy/FeS 2 system is still the most widely used electrode for thermal batteries. However, the performance improvement is limited due to the low thermal stability and low open circuit voltage compared to other materials, and studies have been conducted to supplement these findings. 10-15)
Lee et al. studied the use of carbon black and carbon nanotubes on an FeS 2 cathode in Li-Si/FeS 2 thermal batteries. 10) Discharge measurements and the calculated total polarization confirmed that the performance of the single cell improved when a carbonaceous material was added ( Fig. 2(a, b)). In Fig. 2(c, d), Nyquist plots of data from the FeS 2 cathode are shown for various contents of carbonaceous materials. The impedance of each sample increased as the content increased. The results from a pristine sample were not compared together; therefore, it is difficult to determine what level of impedance the 1 wt% sample represents. In addition, there is no information on how the impedance results were obtained when the content was more than 1 wt%, as well as the discharge and total polarization results when the content was greater than 0.1 wt%. Nevertheless, attempts were made to improve the performance of battery electrodes using carbonaceous materials, 34-43) and the effect was confirmed in the FeS 2 cathode of thermal batteries. However, additional validation is required for actual mass production. Yoon et al. fabricated and evaluated a thin cathode using a tape casting method instead of conventional pellet type cathode. 11) When producing a pellet with a powder compaction process, the thickness is minimized for mechanical strength, thus powder compaction is not an appropriate process from the perspective of optimizing the battery performance. In this study, tape casting was used to improve the utilization of cathode materials and the dispersion condition, binder type, and content of each material were determined. As shown in Fig. 3(a), a thin cathode with maximum content of FeS 2 and a minimum amount of binder was prepared. Its mechanical strength was maintained even under severe deformation, as shown in Fig. 3(b), and its discharge rate was nearly twice that of the pellet type, as shown in the Fig. 3(c, d). Tape casting was used to overcome the limitation of a manufacturing process of an existing pellet and showed excellent performance. Thin cathode production using tape casting was also studied from the perspective of the binder material. Jung et al. fabricated a high thermo-stable poly(imide-co-siloxane) (PIS) binder for thermal batteries and used it with an FeS 2 thin cathode in tape casting. 12) The PIS binder is stable up to 400°C and decomposes at 450°C ( Fig. 4(a)). Thus, it cannot be used at 500°C, which is the operating temperature of normal thermal batteries, but shows a higher level compared to binder materials in typical secondary lithium batteries. 44-48) During discharge, FeS 2 thin cathodes with PIS showed better performance than conventional pellet-type cathodes, and the total polarization was stable. The discharge capacity is lower than the results presented by Yoon et al.. 11) This may arise due to the difference in FeS 2 particle size. The thermal battery is a special cell that operates at high temperature (500°C), thus the active electrode material must have excellent thermal stability up to 500°C. The thermal stability decreases when the particle size of FeS 2 is reduced to a certain size or less, and the decomposition rate increases at 500°C, which decreases the discharge capacity. Another approach to the use of binder materials in thin cathode manufacturing using tape casting is to use multi-walled carbon nanotubes (MWCNTs). Yoon et al. applied MWCNTs, which have excellent mechanical strength and conductivity, as an alternative to conventional organic binders for the thin cathode of thermal batteries. 13) Carboneous materials have been extensively studied and used as additives for electrodes in rechargeable Li-ion batteries. 34-43) Among them, some carboneous materials are used as current collectors and as electrode supports without adding other binder materials. 34,49-54) Noda et al. showed that electrodes containing 99 wt% of active materials and 1 wt% CNTs can be produced. 34) However, more research is required to obtain stable cycle performance without electrode degradation in rechargeable batteries, where charge/discharge cycles repeat. On the other hand, thermal batteries are the primary batteries with only one discharge, and are more suitable than rechargeable batteries in active material-CNT composites as the electrode. That is, the use of an FeS 2-MWCNT composite is a universal method in battery research, but is a suitable and novel method for manufacturing a thin cathode for a thermal battery. Two studies 11,12) related to thin cathode manufacturing with tape casting used organic binders and they were decomposed before reaching the operating temperature of 500°C. The decomposed organic binder material can generate gas that aggravates the performance of the battery, and the residual material can act as a resistor. 55-60) To solve these problems, MWCNTs were used as a binder and conductor for the cathode electrode in this study. MWCNTs have better thermal stability than conventional organic binders and show a mass reduction rate of about 5% at 500°C. Fig. 5(a) shows the contents of each type of cathode. The FeS 2-MWCNT composite cathode has a higher content of FeS 2 than the other types. This is an important factor for increasing the energy density. As shown in Fig. 5(b, c), the FeS 2-MWCNTs composite cathode exhibits higher capacity than the cathode with an organic binder of the same thickness. In this study, MWCNTs are added as a binder in a small amount compared to an organic binder and act as a conductor. All three of the aforementioned studies 11-13) focus on thin cathode manufacturing methods using tape casting. These are new attempts in terms of manufacturing methods to maximize the performance of thermal batteries based on the material properties of FeS 2. That is, it is meaningful to maximize the utilization ratio of the electrode material, which is poor in the production of the conventional pellet-type material, by manufacturing the electrode with the optimum thickness form an electrochemical perspective. Thin cathode manufacturing is possible with tape casting or with any other method capable of producing films with an appropriate thickness, such as spraying 61-65) or screen-printing. 66-70) Rather, the key to thin cathode manufacturing is high performance, stable, highly productive slurry manufacturing technology. 71-74) Of course, thin cathode manufacturing technology is applicable not only to FeS 2 but also to other cathode materials. For this purpose, it is necessary to study the binder, solvent, and dispersant suitable for each cathode material.
3. CoS2 and Bimetallic (Fe-Co) Disulfides
CoS 2 has a lower solubility in molten electrolytes and much higher electronic conductivity, which permits a higher discharge rate. Most importantly, it has a much higher thermal stability and decomposes only above 650°C, which is ~ 100°C higher than for FeS 2, facilitating its long-term application. However, the major disadvantage of CoS 2 relative to FeS 2 is its higher cost. 4) Nevertheless, CoS 2 generally has better properties than FeS 2. For this reason, studies on thermal batteries using CoS 2 cathodes have continued until recently. Zhu et al. fabricated CoS 2 thin cathodes using a screen-printing method and confirmed its feasibility. 16) This study was conducted in the same context as the aforementioned FeS 2 cathode fabricated as a thin film type using tape casting. 11-13) That is, a method for optimizing the thickness in order to maximize utilization of the electrode material was used. A CoS 2 cathode material and screen-printing method were used. Screen-printing is widely used in the textile and electronics industries, and it is a remarkably simple and reproducible production method. In other words, applying this method to thin cathode fabrication for thermal batteries is an appropriate choice. However, this study seems to have a problem in selecting the binder material. In this study, PVDF, which is widely used in the production of electrodes for lithium ion batteries, is used to prepare slurries for screen-printing. However, because the melting point of PVDF is about 160°C, it is not suitable for use in thermal batteries with operating temperatures of approximately 400 to 500°C. As shown in Fig. 6(a), a thin cathode was fabricated with a thickness of 50 μm. As shown in Fig. 6(b), the discharge capacity was more than double that of the pellet-type cathode, producing high voltage and low resistance. However, as described in the aforementioned studies using tape casting, 11-13) it is a natural phenomenon that the utilization rate of the electrode increases to a certain point and the discharge capacity increases when the electrode is thin. In the case of the pellet-type electrode, the thickness of about 500 μm due to the limited mechanical strength, and lost electrode utilization is minimized by adding an electrolyte material. The key points of research for fabricating a thin cathode are 1) material selection (e.g., active material, binder, solvent, and dispersant) and 2) manufacturing method (e.g., tape casting, screen-printing, or spraying). With respect to these two issues, processing methods such as tape casting, 75,76) screen-printing, 77-82) and spraying 83-89) have already been sufficiently investigated in other fields. Of course, appropriate equipment modifications are necessary depending on the application, but this is considered less important than material selection. Very high reliability is required because the thermal battery is a special battery that is primarily used for military applications. Therefore, additional studies on material selection are required to demonstrate the performance while being suitable for the application environment. Liu et al. showed that CoS 2/CNTs composites were prepared using a facile, simple hydrothermal growth process for CoS 2 in the presence of CNTs, as a novel approach of synthesizing cathode composites for thermal battery fabrication. 17) The incorporation of CNTs in the synthesized CoS 2 cathode materials effectively improved the electrical conductivity and discharge performance of the resulting cathode. In this study, single cells with a CoS 2/CNTs cathode, an LiCl-KCl electrolyte, and an Li-Si anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. This study addresses the addition of CNTs to improve the electrochemical properties of pellet-type cathodes, as in the work of Lee et al.. 10) The difference between them is the type of active material and the method used to add CNTs. In this study, CoS 2 was used as a cathode material and a CoS 2/CNT composite was fabricated using a hydrothermal method. Hydrothermal growth was carried out at 180°C by adding CNTs and CoS 2 raw materials to a CNT dispersion. There is no mention of the advantages of hydrothermal reacting raw materials and electrical additive over conventional mixing methods. Intuitively, when the hydrothermal method is used, the interfacial properties of CoS 2 and CNTs improve and the electrical conductivity and discharge performance of the cathode are expected to increase. 90-92) However, in this study, the content is not shown, and the goal is to compare CoS 2/CNTs with CoS 2. Fig. 7(a) shows that the thermal stability of CoS 2/CNTs and CoS 2 are similar. The low thermal stability of CoS 2/CNTs is due to the addition of small amounts of CNTs. CoS 2/CNTs with thermal stability above 600°C can perform better than FeS 2 or FeS 2/additive composites. The discharge evaluation results show that the CoS 2/CNT cathode exhibited excellent performance in all aspects, such as discharge capacity and resistance. However, as in the case of FeS 2, additional validation experiments are required based on these results. Liu et al. also studied carbon-coated CoS 2 that was fabricated using hydrothermal synthesis. 18) In this work, a carbon-coated CoS 2 composite was prepared using a facile onepot hydrothermal method with glucose as a carbon source. During the growth of CoS 2, the glucose molecules were adsorbed and carbonized in situ on the surface of the as-synthesized CoS 2, and the resulting carbon coating provided improved electrical conductivity and discharge performance to the composite. In addition to improving the electrochemical performance, the carbon coating also improves the stability of CoS 2 in air. In this study, single cells composed of a carbon-coated CoS 2 cathode, an LiCl-KCl electrolyte, and an Li-Si anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. The stability of the carbon-coated CoS 2 and bare CoS 2 in air is shown in the XRD and TGA data in Fig. 8(a, b). After 3 months, impure peaks were detected in bare CoS 2, while the carbon-coated CoS 2 remained pure. In addition, bare CoS 2 decomposition occurred before 400°C after 3 months, while the carbon-coated CoS 2 remained relatively stable. In the discharge evaluation results, the carbon-coated CoS 2 cathode showed excellent performance compared to bare CoS 2 in all aspects, such as discharge capacity and resistance. However, as in the case of CoS 2/CNT composites, additional verification tests are required before beginning mass production. Yu et al. used other approaches to improve the stability of CoS 2 in air. In this work, Fe-doped CoS 2 (Co XFe 1−XS 2) compounds were synthesized and evaluated as cathode materials for thermal batteries. 19) Single cells composed of a Co XFe 1−XS 2 cathode, an LiCl-KCl electrolyte, and an Li-Si anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. To investigate the stability of Co XFe 1−XS 2 in air, the as-prepared samples were stored in air (25°C, 100% relative humidity) for 7 days. The XRD patterns Fig. 9(a) do not reveal any apparent impurities or secondary phases in the S-FeS 2 sample, illustrating the stability of FeS 2 in air. Obvious diffraction peaks corresponding to CoSO 4·H 2O can be seen as the content of Co increased further, especially for the S-CoS 2. These results reveal that bimetallic disulfide has better stability in air than that of CoS 2. As shown in Fig. 9(b, c), the bimetallic disulfide sample was superior to CoS 2 in terms of its electrochemical performance, such as its discharge capacity and total polarization. As shown in Fig. 7(d), the discharge capacities of S-CoS 2 and S-Co 0.7Fe 0.3S 2 distinctly decrease due to formation of a large amount of CoSO 4·H 2O, while the S-Co 0.3Fe 0.7S 2 still exhibits good discharge capacity. These results indicate that stability in air has a significant impact on the discharge performance of the disulfides. Bimetallic disulfides with suitable Fe content can exhibit stable discharge performance in air. This approach was also studied by Zhu et al.. 20) This study shows that composite sulfide Fe 0.5Co 0.5S 2 can be prepared through traditional high-temperature solid-state and liquid phase methods, and this composite can be used as a cathode material in thermal batteries. 20) The goal of this study is to improve the poor thermal stability of FeS 2 and the poor stability of CoS 2 in air by applying Fe-Co bimetallic disulfide. In this study, single cells composed of a Fe 0.5Co 0.5S 2 cathode, an LiCl-LiBr-LiF electrolyte, and an Li-Si anode were fabricated and evaluated. The cathode was made of thin film using screen-printing, and other components were fabricated into a pellet-type material using a powder compaction process. PVDF was used as a binder in thin-cathode manufacturing using screen-printing, as in the work of Zhu et al.. 16) In order to manufacture a thin cathode with high reliability, the application of a heat-resistant binder should be considered. Clearly, one can see in Fig. 10(a) that a liquid-phase-synthesized Fe 0.5Co 0.5S 2 sample is purely single-phase while still maintaining a pyrite structure. All diffraction peaks shifted to lower angles than those observed from cubic FeS 2. As shown in Fig. 10(b), Fe 0.5Co 0.5S 2 has better thermal stability compared to FeS 2. Better thermal stability points to a higher utilization rate for electrode materials, which can greatly increase the discharge performance. Fig. 10(c, d) shows the morphology of Fe 0.5Co 0.5S 2 prepared using solid-state and liquid phase methods. The particle size of the liquid-phase-synthesized Fe 0.5Co 0.5S 2 is much smaller, which is expected to have a significant impact on discharge performance. In other words, larger specific surface area can greatly decrease electrochemical polarization, thus significantly slowing down the working voltage drop and improving discharge capacity. The discharge evaluation results in Fig. 10(e, f) show this tendency. Fe0.5Co0.5S2 cathodes have higher discharge capacity than an FeS2 cathode, which is due to the improved thermal stability provided by bimetallic Fe-Co. In addition, liquid phase synthesized Fe0.5Co0.5S2 has a higher discharge capacity than solid-state synthesized Fe0.5Co0.5S2. This is primarily due to the pure single-phase and small particle size of the aforementioned liquid phase synthesized Fe0.5Co0.5S2. New approaches to take advantage of each substance, not limited to conventional disulfide cathodes, are very encouraging.
4. NiS2
The thermal stability of NiS 2 is intermediate between that of FeS 2 and CoS 2, as shown in Fig. 11(a). 4) The electrochemical performance of NiS 2 in an LiCl-KCl eutectic electrolyte is compared to that of synthetic FeS 2 and CoS 2 in Fig. 11(b). As expected, the voltage response for the cell with the NiS 2 cathode was intermediate between that of the CoS 2 and FeS 2 cells. However, the overall performance of the NiS 2 cathode was similar to that of CoS 2. Because the costs of NiS 2 precursors are much less than those for CoS 2, the similar electrochemical performance motivates the use of NiS 2 for application where CoS 2 might have been used. 1-4) However, NiS 2 does not provide a cost advantage over FeS 2 and is not attractive enough to offset the high cost in terms of performance. Several studies that expand on this point have been published. To maximize the performance of NiS 2, Zhou et al. fabricated NiS 2 as a nanostructure and evaluated its properties and performance. 22) In this study, single cells composed of a nano-NiS 2 cathode, an LiCl-LiBr-LiF electrolyte, and an Li- B anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. Nanostructured NiS 2 powders were prepared by ball-milling for 10 to 40 h and subsequently heating at 400°C for 1 h using carbonyl nickel and sulfur as precursors. The theoretical capacity of NiS 2 is about 870 mAh/g, but the discharge capacity in practical applications is much lower. The sluggish diffusivity of lithium ions in the NiS 2 cathode and low surface area of the bulk electrodes are identified as the primary factors producing poor discharge performance. 93) In addition, intermediate phase evolution with low conductivity can reduce the discharge performance. 94,95) In this study, the ball-milling time was defined as a control parameter to characterize the particle size of NiS 2. As shown in Fig. 12(b), the surface area of the 40 h ball-milled NiS 2 is the largest, which can increase the contact area of the electrode and the electrolyte, thereby improving the discharge performance. The discharge capacity of 40 h ball-milled NiS 2 was largest at the discharge curves (0.1 A/cm 2) shown in Fig. 12(c). The discharge curves (0.5 A/cm 2) in Fig. 12(d) show the same tendency in the discharge capacity, but the voltage plateau becomes clearer. This is due to the intermediate phase generated during discharge, and it becomes clearer as the size of NiS 2 particles decreases. That is, if the size of NiS 2 particles is large, the reaction mechanism of NiS 2 does not occur completely. Inferred from the results of this study, the negative influence of the intermediate phase (NiS) becomes stronger when the NiS 2 particles are larger due to the relatively low conductivity of NiS, 96) leading to a decrease in discharge performance. In other words, NiS 2 produced rapid intermediate phase evolution, which increased the discharge capacity of the NiS 2 cathode. This mechanism is shown in Fig. 12(a). This study is meaningful for improving the characteristics of the NiS 2 cathode and for interpreting the performance improvement mechanism in various ways. $$$Zhou et al. studied carbon-modified nano-NiS 2 based on the aforementioned nano-NiS 2 results. 23) In this study, single cells composed of a carbon-coated nano-NiS 2/nano-NiS 2 cathode, an LiCl-LiBr-LiF electrolyte, and an Li-B anode were fabricated and evaluated. All components were fabricated into a pellet-type material using a powder compaction process. Carbon modification is always seen as an effective method for improving the conductivity and thermal/structural stability of electrodes. 97-102) In this study, NiS 2 particles were coated with amorphous carbon. Then they accumulated into submicron particles and were connected/fixed by a carbon network. These effects are shown in Fig. 13(e, f), as shown in the TEM image and schematic diagram. The initial decomposition temperature of nano-NiS 2 increased from 400 to 590°C after carbon modification. As shown in Fig. 13(b), the discharge performance improves as the thermal stability increases. In the case of carbon-coated nano-NiS 2, the discharge capacity at high temperature shows a stable specific capacity up to 600°C near the initial decomposition temperature ( Fig. 13(c)). Beyond 600°C, NiS 2 rapidly decomposes and its specific capacity decreases. Carbon modification not only improves the conductivity of the cathode on its own, but it also inhibits the spread of the low conductivity product Li 2S. This effect is reflected in the pulse discharge and resistance results ( Fig. 13(d)).
5. NiCl2
Due to several advantages mentioned above, disulfide-based materials have been used as the cathode in thermal batteries for decades. However, it is difficult for disulfide materials to meet recent demands for high power thermal batteries due to their low thermal stability and low discharge voltage. As a result, research and development on cathode materials (e.g., NiCl 2) other than disulfide-based materials have continued. NiCl 2 has been considered as one promising cathode material for use in thermal batteries thanks to its high relative potential, high current discharge, high specific energy, and low cost. However, its high solubility in molten salt electrolytes causes thermal batteries to short circuit, and its low electronic conductivity results in long activation time, thus NiCl 2 thermal batteries cannot provide electrochemical performance. 25)
Zhou et al. conducted surface modification with a carbon coating to improve the performance of NiCl 2-based batteries. 25) It was confirmed that modification using carbon coating has a positive effect on electrochemical performance of CoS 218) and NiS 2. 23) In this study, the solubility of NiCl 2 in the molten salt was evaluated using a contact angle measurement on the NiCl 2 cathode, which could be used to analyze wetting of the LiCl-LiBr-LiF electrolyte. These results indicate that the carbon coating could inhibit NiCl 2 from dissolving into the molten salt ( Fig. 14(a)). Single cells composed of carbon-coated nano-NiCl 2/pure NiCl 2 cathodes, an LiCl-LiBr-LiF electrolyte, and an Li-B anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. The carbon-coated NiCl 2 thermal batteries exhibited increased specific capacity and increased specific energy compared to that of pure NiCl 2 thermal batteries. Zhu et al. investigated an alternative to the conventional stainless steel substrate for NiCl 2-based thermal batteries. 26) In this study, an NiCl 2 cathode was fabricated by screen printing on stainless steel and nickel foam substrates. In addition, LiCl-LiBr-LiF electrolyte and Li-Si alloy anodes were fabricated into pellet-type materials using a powder compaction process. The shape of the NiCl 2 cathode in Fig. 15(a, b) shows that NiCl 2 penetrates the Ni foam. In Fig. 15(b), NiCl 2 does not penetrate to the back of the Ni foam, but this could be improved by adjusting the viscosity of the NiCl 2 slurry and the screen-printing processing conditions. Furthermore, additional research on Ni foam may be needed because the porosity of the Ni foam and the shape of the pore can greatly affect the performance of the cathode. The peak voltage of the NiCl 2 cathode using an Ni foam substrate is about 2.55 V, which is much higher than that of sulfide cathode materials. In addition, the NiCl 2 cathode with a Ni foam substrate has a specific capacity of 684.61 Asg −1 with a cut-off voltage of 1.5 V (stainless steel substrate: 299.39 Asg −1). This improvement can be attributed to the lower resistance of the NiCl 2 cathode on the Ni foam substrate ( Fig. 15(d)). It is significant that the results from this study can be used to improve the mass productivity of NiCl 2 cathodes by using a metal foam substrate, which can greatly improve the electrochemical performance and screen printing process, making them suitable for mass production. This approach seems to be applicable to other cathode materials besides NiCl 2, and it is important to consider the selection of suitable metal foams and shapes. In addition, because metal foam is expected to act as a support for the cathode material, it is also necessary to minimize the amount of binder material added to the slurry.
6. Copper Vanadium Oxide
Copper vanadium oxide provides high voltage, high capacity, and high thermal stability. V 2O 5 and CuO were previously evaluated for use as possible cathodes in thermal batteries. 29) In addition, the electrochemistry of Cu 5V 2O 10 was reported by Sakurai et al. 103) and Eguchi et al.. 104)
A series of the CuO-V 2O 5 oxides, such as CuV 2O 6, Cu 2V 2O 7, and Cu 5V 2O 10 were synthesized and evaluated as possible candidate cathode materials in high voltage thermal batteries. 29) In this study, single cells composed of a copper vanadium oxide cathode, an LiCl-KCl electrolyte, and an Li-Al anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. In the Fig. 16(a), Cu 2V 2O 7 showed lower thermal stability than CuV 2O 6, but the weight loss was negligible (0.085%) up to 600°C. This is much smaller than the weight loss observed with sulfide cathode materials. A 3.4 V voltage plateau at a current density of 100 mA/g and a 2.5 V voltage plateau at a current density of 310 mA/g were observed during discharge at 525°C. This is a much higher value than was observed with FeS 2, which has a voltage plateau ranging from 1.5 to 2 V under similar conditions, and is a characteristic that can contribute to increasing the energy density. Dai et al. studied Cu 3V 2O 8 and Cu 5V 2O 10 thin-film cathodes for thermal batteries. 30) In this study, single cells composed of a copper vanadium oxide cathode, a high-voltage compatible electrolyte, and an Li-Si anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. The cathode was also fabricated as a thin film using tape casting in order to facilitate a suitable performance comparison. The high-voltage compatible electrolyte consists of LiPO 3, Li 2SO 4, Li 2CO 3, and LiF. Copper vanadium oxide has higher voltage than sulfide materials; therefore, it is necessary to study the molten salt electrolyte within a suitable voltage window. 105) In this study, the final constant stable open circuit voltage (OCV) is 2.971 V at 500°C. The discharge profile of the Cu 5V 2O 10 thin films contains multiple plateaus, similar to what was observed with a pressed film ( Fig. 17(a)). The discharge profile of Cu 3V 2O 8 shows a much more obscure phase change than that of Cu 5V 2O 10. To determine the cause of these differences, an analysis of the discharge reaction for each substance would be necessary. The specific capacity of a thin film cathode is higher than that of a pressed cathode, regardless of the cathode materials. Copper vanadium oxide has excellent properties, such as high voltage and high thermal stability, but studies on this material remain lacking. Applying the functional coating or manufacturing method mentioned above and optimizing the material properties were may significantly contribute to the development of thermal batteries with high energy density.
7. Other Materials
In addition to the aforementioned materials, other cathode materials for thermal batteries have been studied recently. Irvine et al. investigated ZrS 3 as a possible cathode material for use in thermal batteries. 31 ZrS 3 has a pseudo one-dimensional structure ( Fig. 18(a)). In this study, single cells composed of a ZrS 3 cathode, an LiCl-KCl electrolyte, and an Li-Si anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. A single 1.7 V voltage plateau at a current density of 11 mA/cm 2 was observed during discharge at 500°C with a capacity of 357 mAh/g. However, these results are inferior to existing sulfide materials, such as FeS 2, CoS 2 and NiS 2. One thing to note is the high thermal stability of 700°C. This study is an early attempt to apply ZrS 3 as the cathode in thermal batteries, thus additional work should focus on improving the performance of these devices. Irvine et al. investigated CoNi 2S 4 as a candidate cathode material for use in thermal batteries. 32) CoNi 2S 4 adopts an inverse spinel structure, as shown in Fig. 18(d). In this study, single cells composed of a CoNi 2S 4 cathode, an LiCl-KCl electrolyte, and an Li-Si anode were fabricated and evaluated. All components were fabricated into pellet-type materials using a powder compaction process. The CoNi 2S 4 cell exhibits two voltage plateaus at 500°C, one at 1.75 V and the second at 1.5 V. The CoNi 2S 4 cell has a specific capacity of 318 mAh/g from 2.58 to 1.25 V OCV. However, these results are inferior to those mentioned above. CoNi 2S 4 shows the thermal stability of 550°C, which is lower than that of FeS 2; hence, its application scope becomes even narrower. It is encouraging that the development of new cathode materials for thermal batteries is ongoing. However, future studies should focus on improving the capacity, thermal stability, and voltage currently required in industry.
8. Conclusions
Thermal batteries are primarily limited to military applications because of their specificity. This closed and specific propensity is reflected in research activities, and the number of published studies is very small compared to studies on general Li ion batteries. Nevertheless, over the past decade, much valuable research has been conducted on cathode materials for thermal batteries. Table 1 summarizes the main results of recent studies. Today, electronics industries in all fields are rapidly developing, and demand for power sources with high energy density is increasing. The military requires a high-performance power source that can power advanced weapons systems. In other words, future thermal batteries require high energy density and high reliability against external stimuli. High capacity, high voltage, and high thermal stability are the three most important considerations for the development of high performance thermal batteries. There are four major approaches to improve these.
Surface modification: Coating with carbon or other materials to improve the thermal stability or electrical conductivity of conventional cathode materials. Alloy and compound: Synthesis of alloy or compound materials to improve the thermal stability or specific capacity of conventional cathode materials. Manufacturing method: Manufacturing of a thin film cathode to overcome the limitations of pellet-type materials formed via powder compaction. New materials
It would be possible to develop a cathode for high performance thermal batteries by appropriately combining the above considerations.
Acknowledgments
This research was supported by ‘Agency for Defense Development’ (2019-0060) as a collaborative core technology research project.
Fig. 1
Cross-sectional view of an Li-Si/FeS 2 thermal battery. Reprinted with permission from Ref. [ 1]. Copyright 2006 Elsevier B. V. 
Fig. 2
(a) Single cell discharge results and (b) total polarization values of pristine, 0.1 wt.% CB, and 0.1 wt.% MWCNT samples. (c, d) Nyquist plots for CB-added and MWCNT-added samples. Reprinted with permission from Ref. [ 10]. Copyright 2013 The Korean Society of Industrial and Engineering Chemistry. 
Fig. 3
(a) The contents of substances contained in the thin cathode and pellet type cathode. (b) Photograph of a thin cathode prepared using tape casting. (c) Discharge graphs of the thin cathode and pellet-type cathode. (d) Discharge results of the thin cathode and pellet-type cathode. Reprinted with permission from Ref. [ 11]. Copyright 2017 Elsevier Ltd and Techna Group S.r.l. 
Fig. 4
(a) Schematic view of the FeS 2/poly(imide-co-siloxane) composite cathode. (b, c) Discharge capacity and total polarization of the FeS 2/PIS thin cathode and pellet type cathode. (d) TGA data. (e, f) SEM images of the thin cathode and pellet-type cathode after 1 h treatment at 450°C. Reprinted with permission from Ref. [ 12]. Copyright: This is an open access article distributed under the Creative Commons Attribution License. 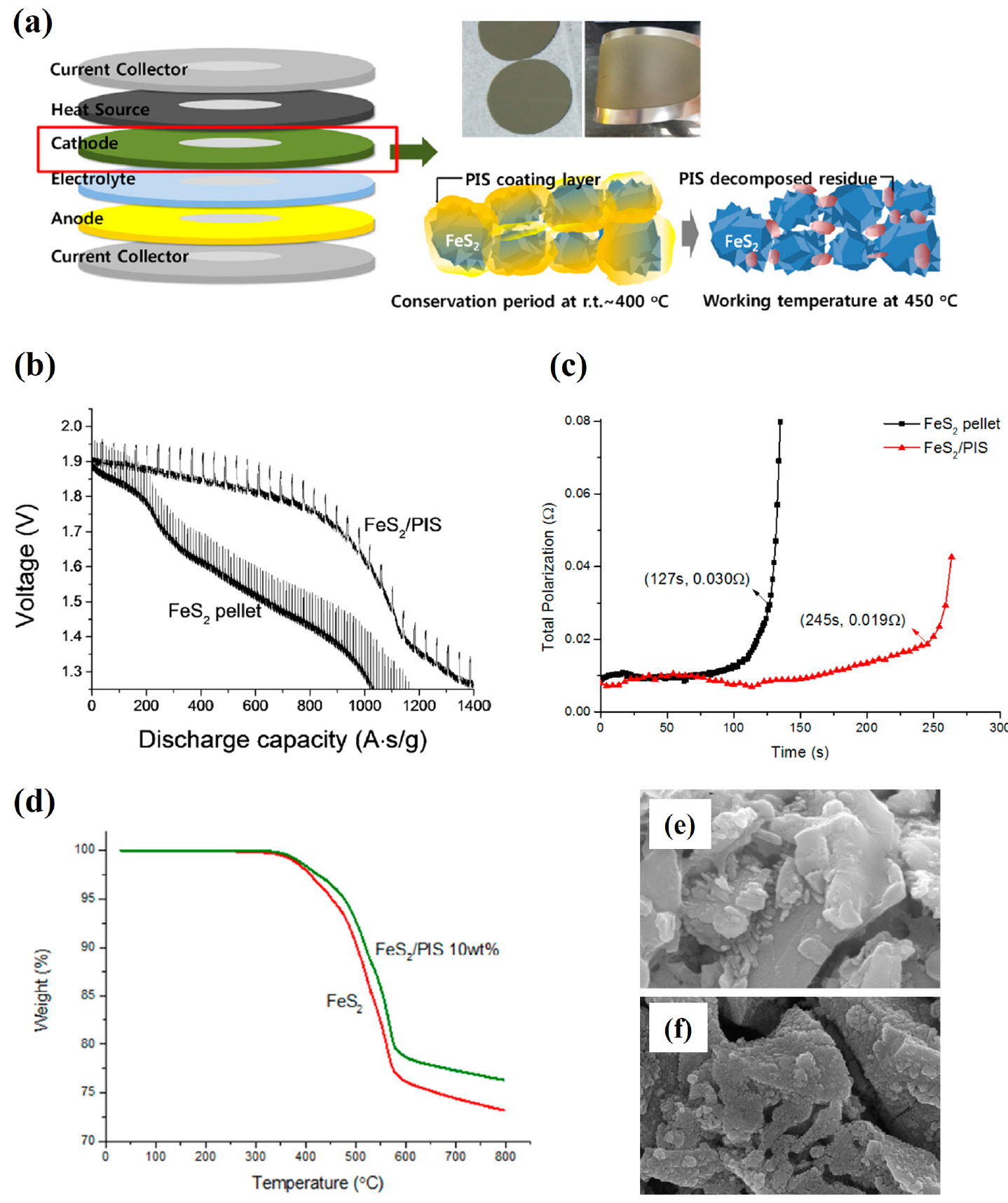
Fig. 5
(a) Content of substances contained in the cathodes. (b) Discharge graphs of the cathodes. (c) Discharge results of the cathodes. Reprinted with permission from Ref. [ 13]. Copyright 2017 The American Ceramic Society. 
Fig. 6
(a) Photograph and SEM images of a thin film cathode prepared using screen-printing. (b) Discharge curves and (c) resistance changes of the thin cathode and pellet cathode. Reprinted with permission from Ref. [ 16]. Copyright 2017 Elsevier B. V. 
Fig. 7
(a) Thermal stability of CoS 2 and CoS 2/CNTs. (b) SEM image of CoS 2/CNTs. (c) Specific discharge capacity, (d) energy, (e) pulse power, and (f) polarization data from CoS 2 and CoS 2/CNTs. Reprinted with permission from Ref. [ 17]. Copyright 2016 Elsevier Ltd. 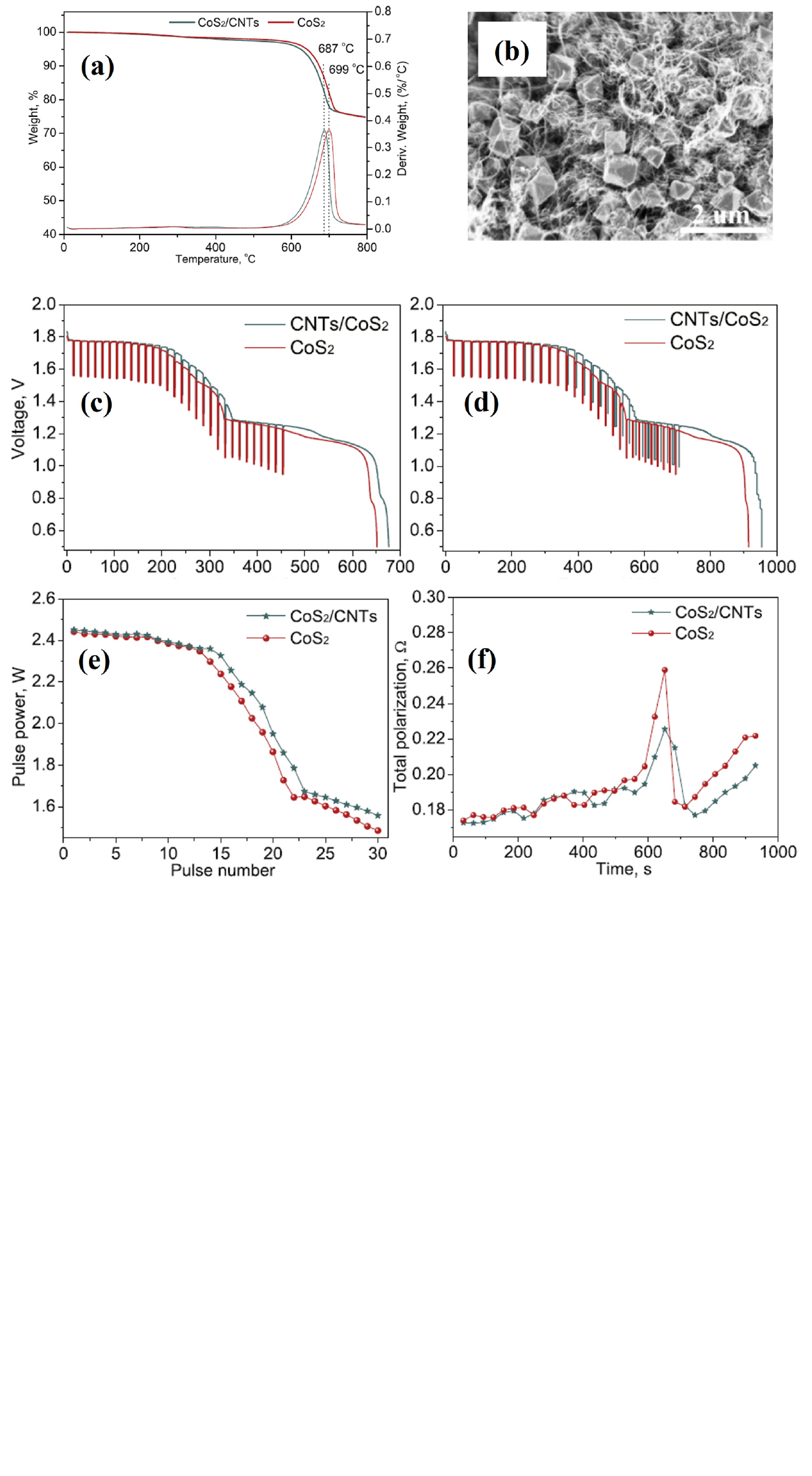
Fig. 8
(a) XRD patterns of fresh products and products that were stored for 3 months. (b) TG curves of CoS 2 and C@CoS 2 after being stored in dry air for 3 months. (c) Discharge capacity, (d) energy, (e) pulse power, and (f) polarization data from CoS 2 and C@CoS 2. Reprinted with permission from Ref. [ 18]. Copyright 2017 published by Elsevier Ltd. 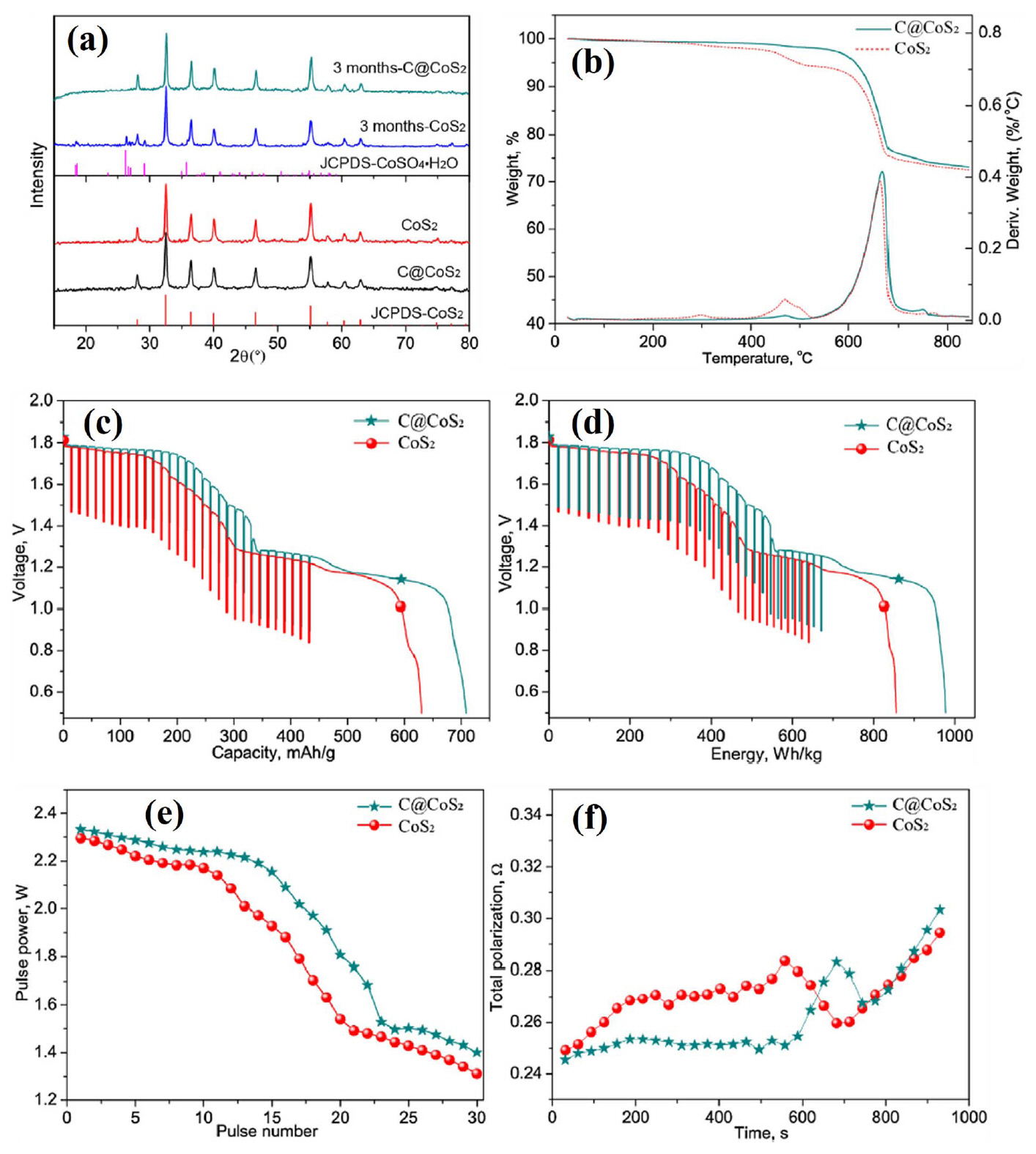
Fig. 9
(a) XRD patterns of S-Co XFe 1−XS 2 (samples stored in air). (b) Discharge performance and (c) resistance of the Co XFe 1−XS 2 cell. (d) Discharge performance of the S-Co XFe 1−XS 2 cell. Reprinted with permission from Ref. [ 19]. Copyright: This is an open access article distributed under the Creative Commons Attribution License. 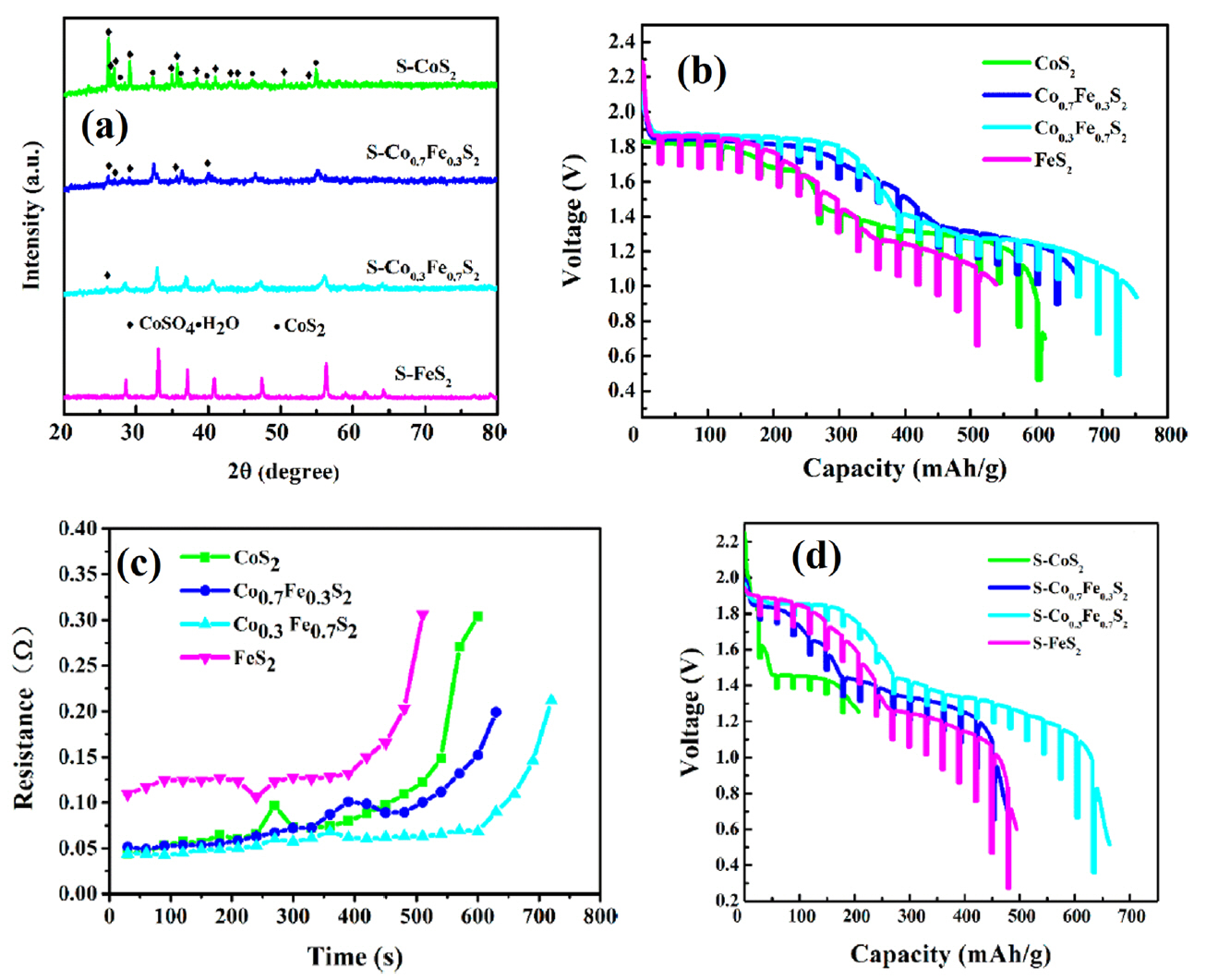
Fig. 10
(a) XRD patterns on Fe 0.5Co 0.5S 2 synthesized using two different methods. (b) Thermal analysis curves. SEM images of (c) S-Fe 0.5Co 0.5S 2 and (d) L-Fe 0.5Co 0.5S 2. (e) Discharge curves for single cells. (f) Pulsed discharge curves and resistance change curves of single cells. Reprinted with permission from Ref. [ 20]. Copyright 2018 Elsevier B. V. 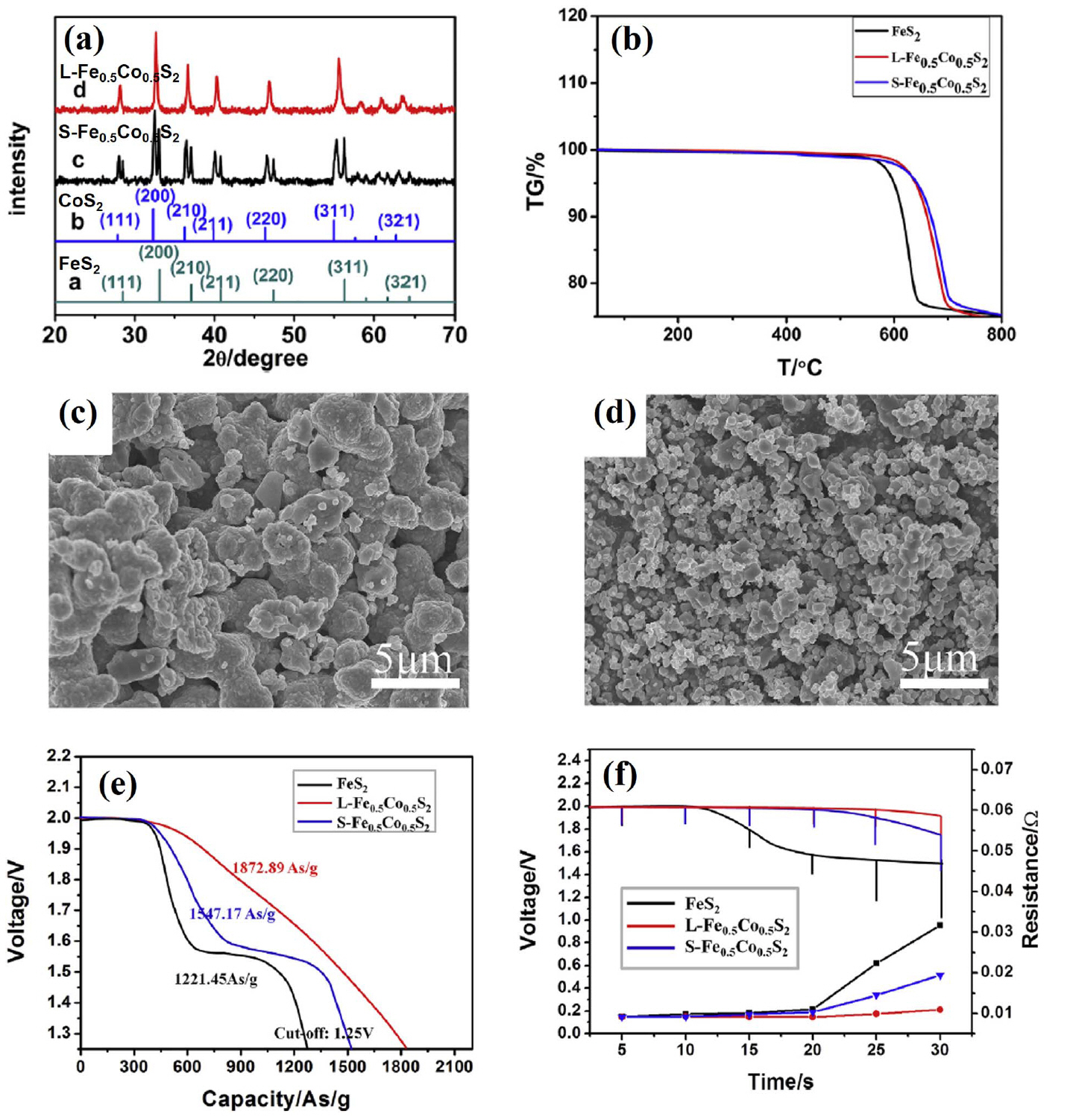
Fig. 11
(a) TGA traces for synthetic FeS 2, CoS 2, and NiS 2 in argon. (b) Discharge of Li-Si/LiBr-KBr-LiCl/MS 2 cells at 500°C. Reprinted with permission from Ref. [ 4]. Copyright 2007 Elsevier B. V. 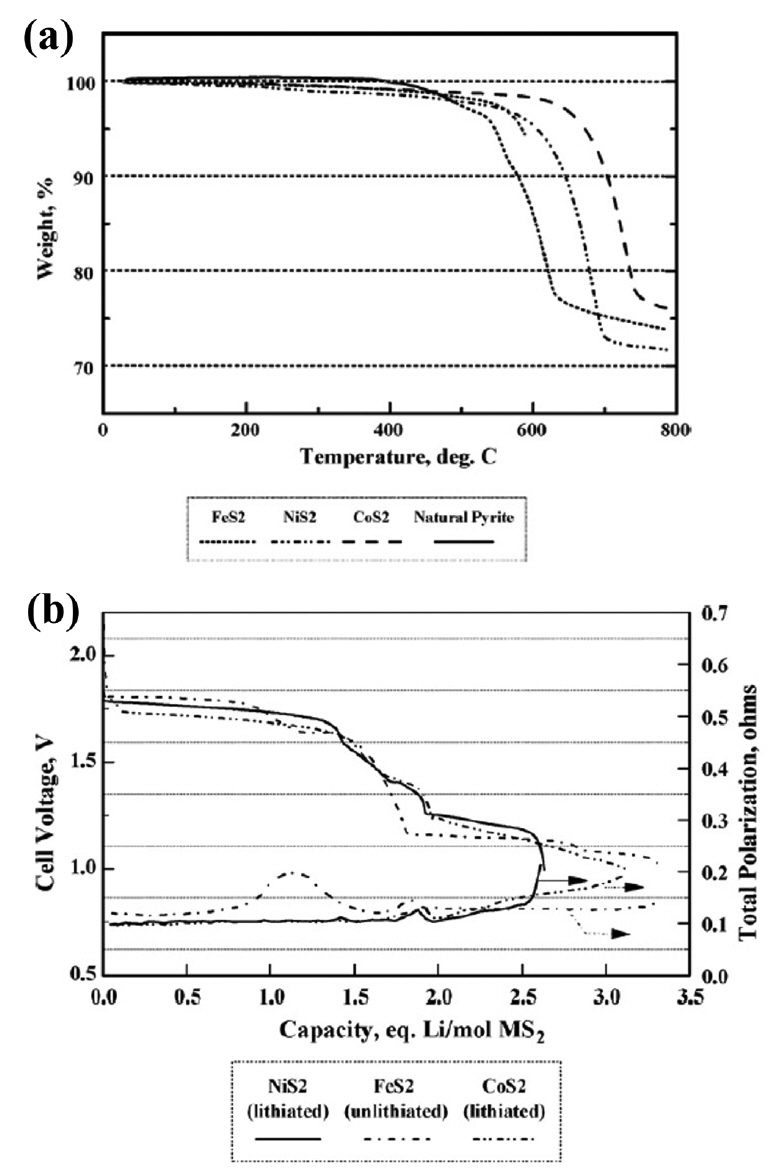
Fig. 12
(a) The effect of nano-NiS 2 in a thermal battery. (b) N 2 adsorption-desorption isotherms (inset: BET surface area). Discharge performance of NiS 2 with different ball-milling times at current densities of (c) 0.1 A/cm 2 and (d) 0.5 A/cm 2. (e) pulsed discharge performance. (f) Resistance variation. Reprinted with permission from Ref. [ 22]. Copyright 2017 Elsevier B. V. 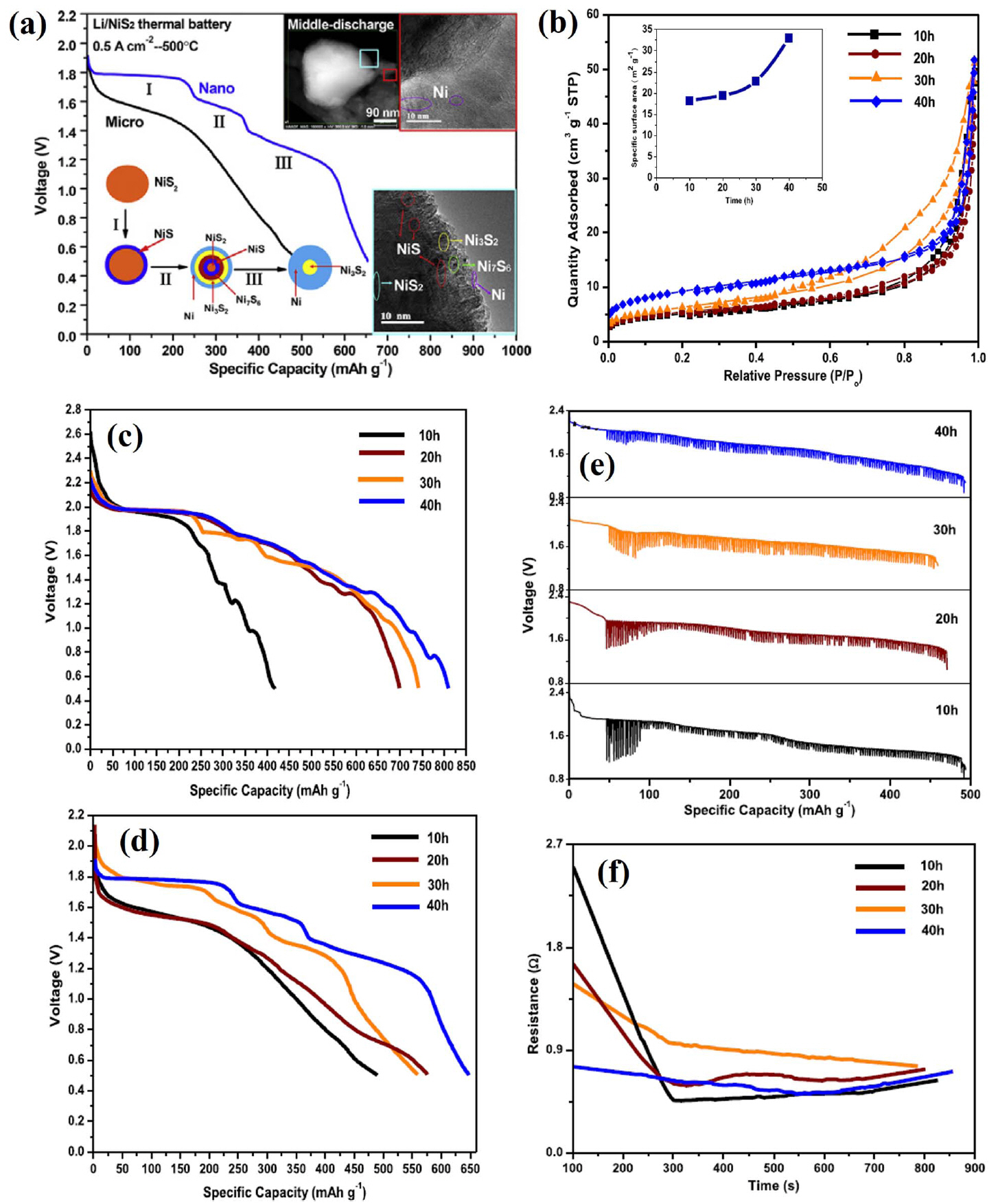
Fig. 13
(a) TG data from nano-NiS 2 before and after carbon modification. (b-c) Specific capacities of NiS 2 and NiS 2/C at 0.1 A/cm 2. (d) Pulse discharge and resistances. (e) TEM images of discharge products. (f) Schematic diagram showing carbon modification. Reprinted with permission from Ref. [ 23]. Copyright 2018 The Royal Society of Chemistry. 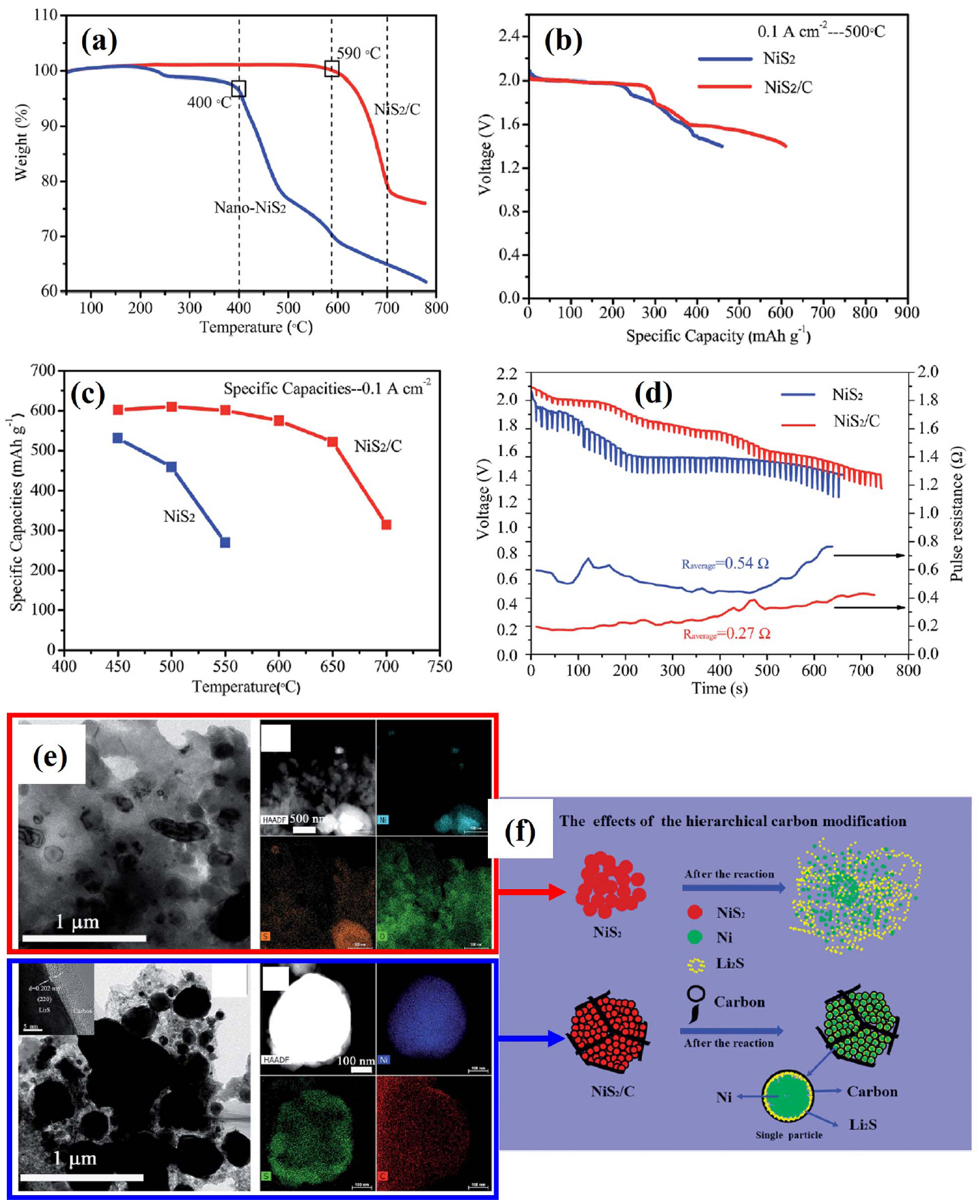
Fig. 14
(a) Contact angle test. (b-d) Comparison of the electrochemical performance of single-cell thermal batteries with carbon-coated NiCl 2 and pure NiCl 2 cathodes at 480°C and at various current densities. Reprinted with permission from Ref. [ 25]. Copyright 2017 Elsevier B. V. 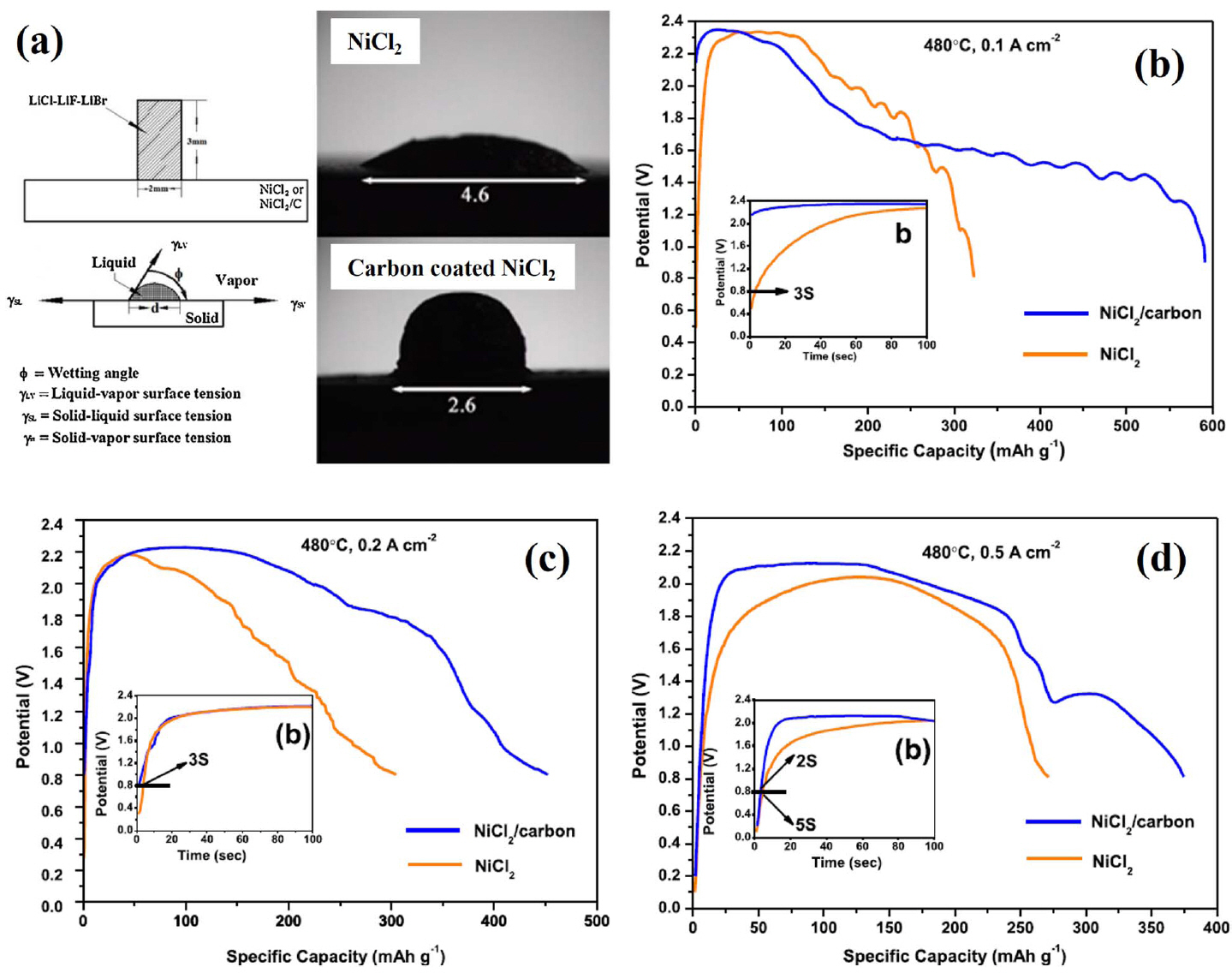
Fig. 15
(a) Photographs of the NiCl 2 cathode. (b) Cross-sectional SEM images of the NiCl 2 cathode. (c) Discharge curves and (d) resistance changes of single cells with NiCl 2 cathodes. Reprinted with permission from Ref. [ 26]. Copyright 2017 Elsevier B. V. 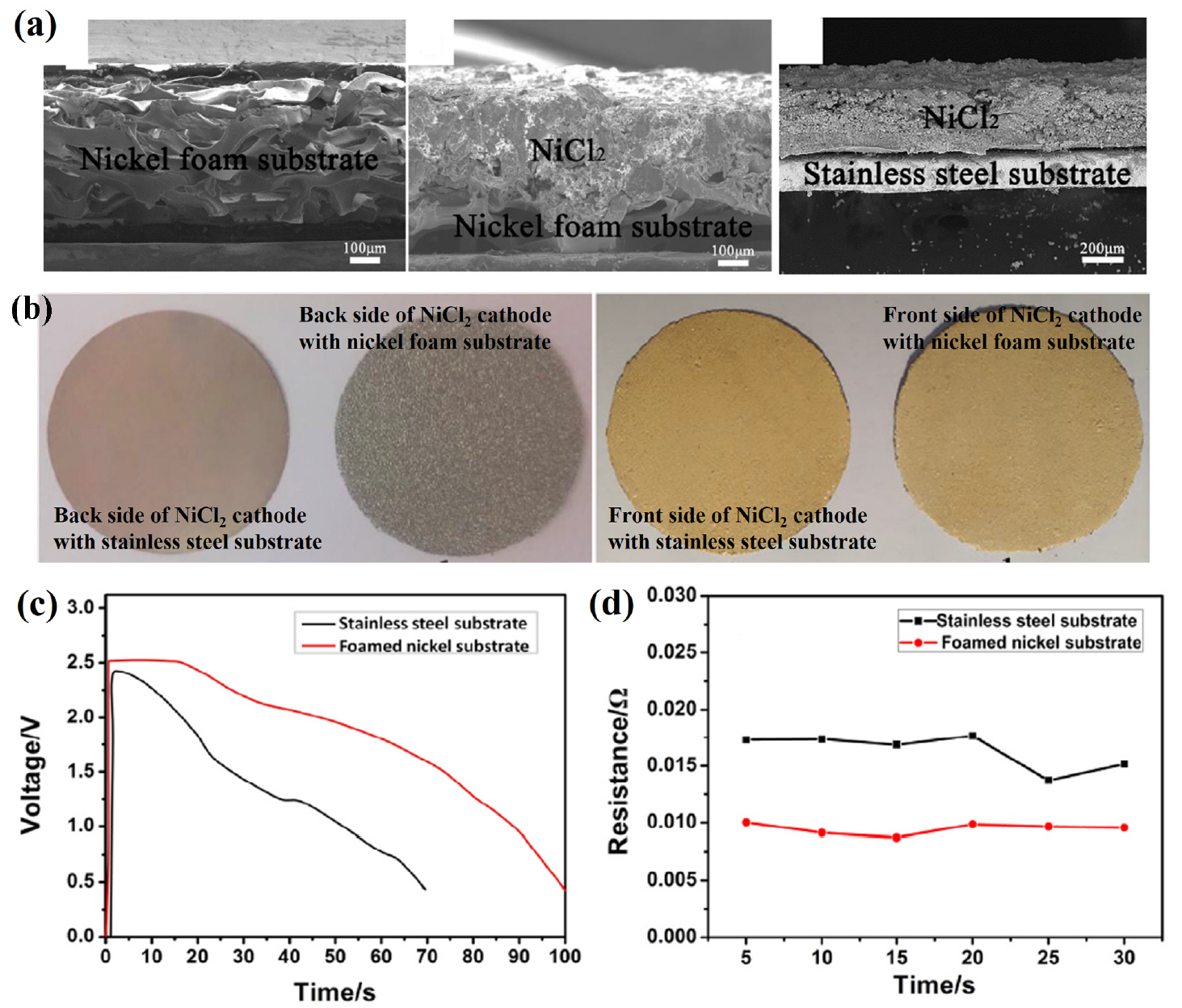
Fig. 16
(a) TGA curves of CuV 2O 6 and Cu 2V 2O 7. Discharge curves of the (b) CuV 2O 6 and (c) Cu 2V 2O 7 cathodes. Reprinted with permission from Ref. [ 29]. Copyright 2012 Elsevier B. V. 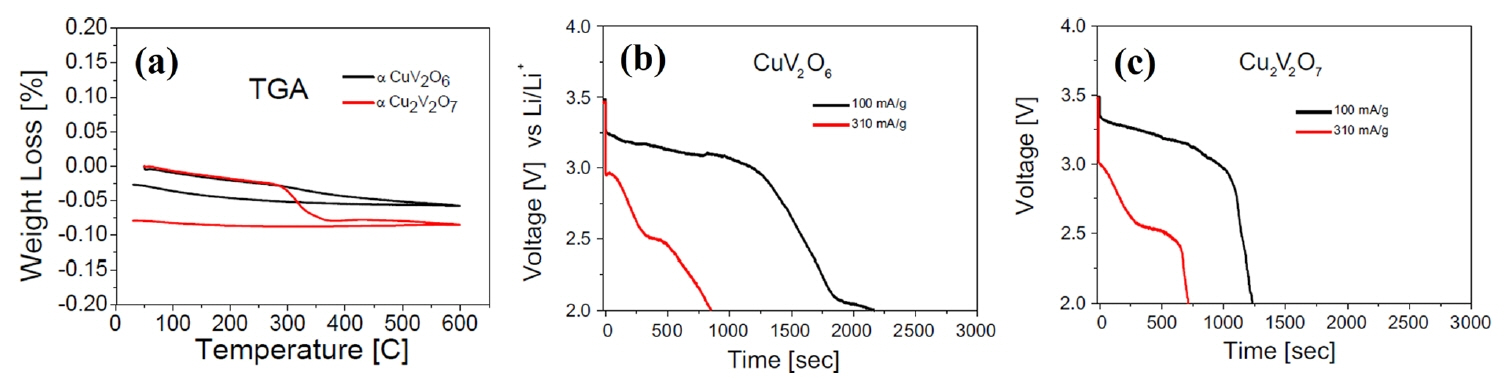
Fig. 17
(a) Discharge profile for an LiSi/Cu 5V 2O 10 cell. (b) Polarization of an LiSi/Cu 5V 2O 10 cell. (c) Discharge profile of an LiSi/Cu 3V 2O 8 cell. (d) Polarization of an LiSi/Cu 3V 2O 8 cell. Reprinted with permission from Ref. [ 30]. Copyright 2011 The Electrochemical Society. 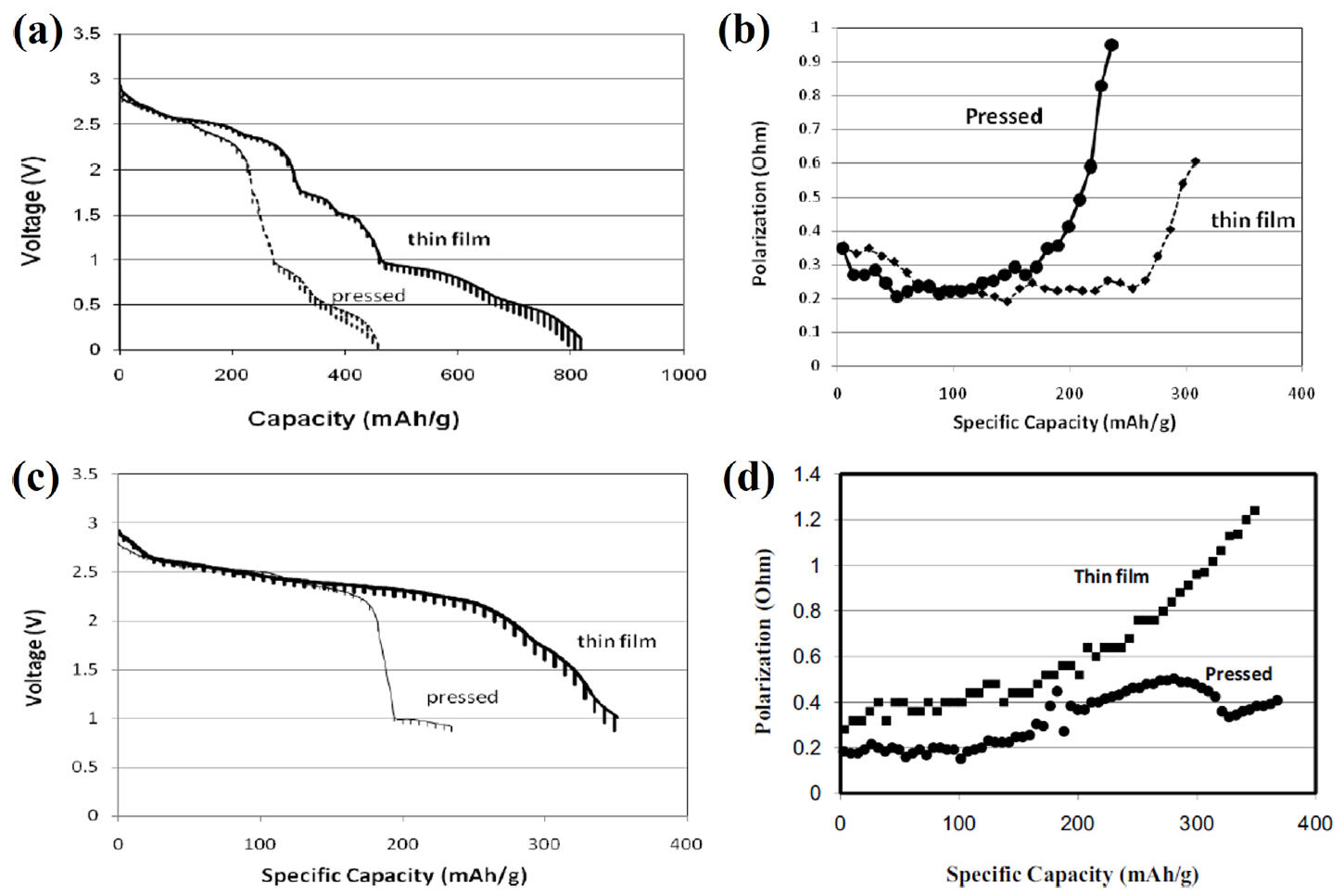
Fig. 18
(a) Crystal structure of ZrS 3. (b) SEM image of a ZrS 3 cathode. (c) Discharge performance of an LiSi/ZrS 3 cell. Reprinted with permission from Ref. [ 31]. Copyright: This is an open access article distributed under the Creative Commons Attribution License. (d) Crystal structure of CoNi 2S 4. (e) SEM image of CoNi 2S 4. (f) Discharge performance of an LiSi/CoNi 2S 4 cell. Reprinted with permission from Ref. [ 32]. Copyright: This is an open access article distributed under the Creative Commons Attribution License. 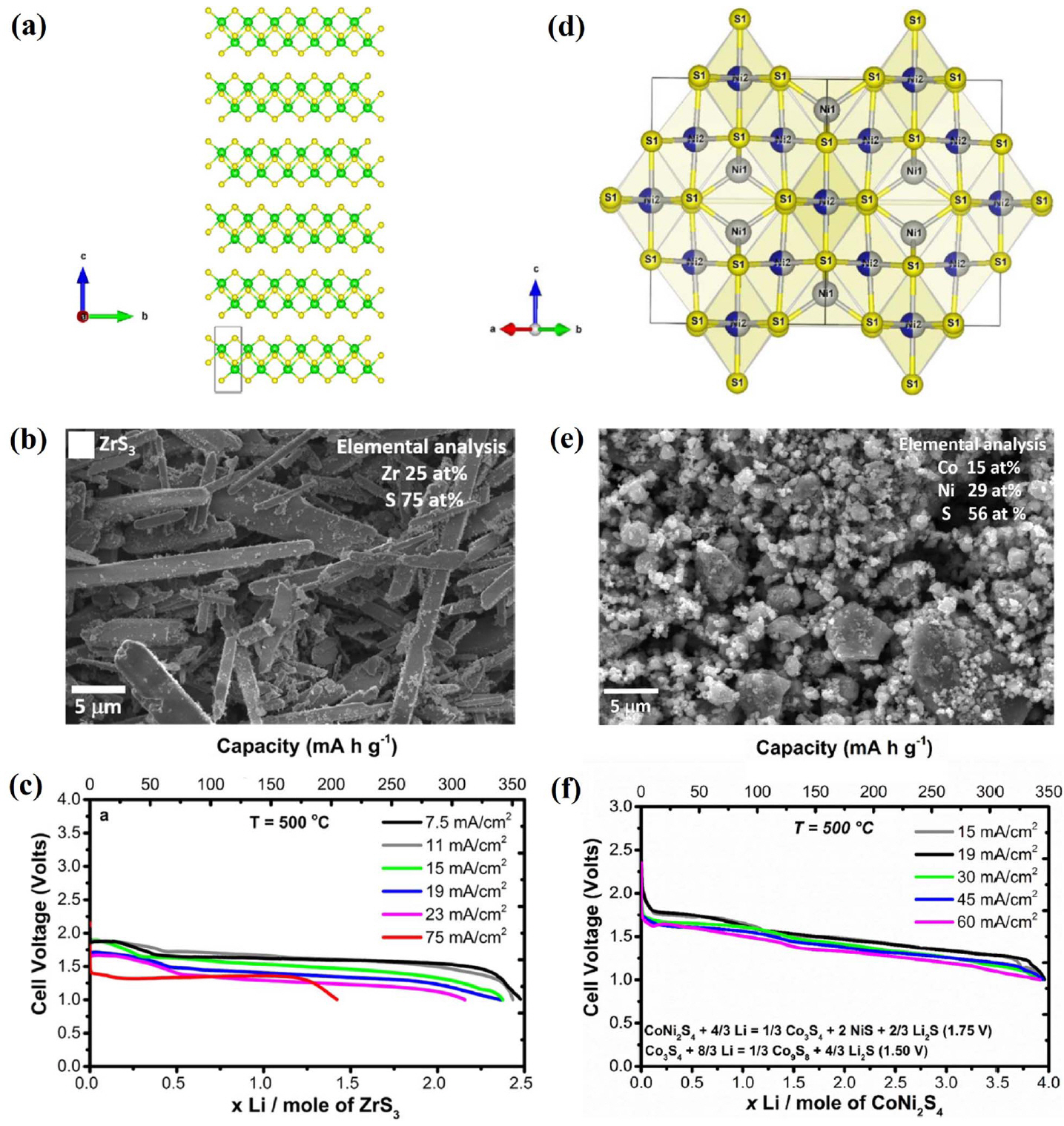
Table 1
Summary of Recent Studies on Cathode Materials for Thermal Batteries
|
Cathode |
Anode |
Electrolyte |
Cathode type/Thickness (μm) |
Specific capacity(A ·sec/g) |
Discharge temperature (°C) |
Cut-off (V) |
OCV (V) |
Voltage plateau (V) |
Thermal stability (°C) |
Ref. |
|
1 |
FeS2
|
Li-Si |
LiCl-KCl |
Pellet/ - |
- |
500 |
1.56 |
1.9 |
1.75/ 1.5/ 1.1 |
580 |
[10] |
|
2 |
FeS2
|
Li-Si |
LiCl-KCl |
Thin film/ 100 |
1934 |
500 |
1.3 |
2.0 |
1.75/ 1.4 |
580 |
[11] |
|
3 |
FeS2
|
Li-Si |
LiCl-KCl |
Thin film/ 100 |
1283 |
500 |
1.3 |
1.9 |
1.8/ 1.4 |
580 |
[12] |
|
4 |
FeS2
|
Li-Si |
LiCl-KCl |
Thin film/ 50 |
2305 |
500 |
1.3 |
2.0 |
1.85/ 1.4 |
580 |
[13] |
|
5 |
CoS2
|
Li-Si |
LiCl-LiBr-LiF |
Thin film/ 50 |
2092 |
500 |
1.25 |
1.99 |
1.9/ 1.76 |
650 |
[16] |
|
6 |
CoS2
|
Li-Si |
LiCl-KCl |
Pellet/ - |
791.28 |
500 |
1.7 |
1.84 |
1.77/ 1.3 |
650 |
[17] |
|
7 |
CoS2
|
Li-Si |
LiCl-KCl |
Pellet/ - |
848.88 |
500 |
1.7 |
1.84 |
1.78/ 1.3 |
650 |
[18] |
|
8 |
Co0.3Fe0.7S2
|
Li-Si |
LiCl-KCl |
Pellet/ - |
2304 |
525 |
1.0 |
2 |
1.85/ 1.3 |
620 |
[19] |
|
9 |
Fe0.5Co0.5S2
|
Li-Si |
LiCl-LiBr-LiF |
Thin film/ 50 |
1872.89 |
- |
1.25 |
2.04 |
2 |
620 |
[20] |
|
10 |
NiS2
|
Li-B |
LiCl-LiBr-LiF |
Pellet/ - |
2858.4 |
500 |
0.5 |
2.2 |
1.98/ 1.8/ 1.4 |
400 |
[22] |
|
11 |
NiS2
|
Li-B |
LiCl-LiBr-LiF |
Pellet/ - |
2196 |
500 |
1.4 |
2.0 |
2.0/ 1.6 |
590 |
[23] |
|
12 |
NiCl2
|
Li-B |
LiCl-LiBr-LiF |
Pellet/ - |
2127.6 |
480 |
0.8 |
2.35 |
2.3/ 1.5 |
- |
[25] |
|
13 |
NiCl2
|
Li-Si |
LiCl-LiBr-LiF |
Nickel foam/400 |
684.61 |
- |
1.5 |
2.55 |
- |
- |
[26] |
|
14 |
CuV2O6
|
Li-Al |
LiCl-KCl |
Pellet/ 1000 |
220 |
525 |
2.0 |
3.5 |
- |
600 |
[29] |
|
15 |
Cu2V2O7
|
Li-Al |
LiCl-KCl |
Pellet/ 1000 |
125 |
525 |
2.0 |
3.5 |
- |
600 |
[29] |
|
16 |
Cu5V2O10
|
Li-Si |
LiPO3, LiSO4, Li2CO3, LiF |
Thin film/ 80 |
1044 |
500 |
2.0 |
2.971 |
2.5/ 1.75/ 1.5 |
- |
[30] |
|
17 |
Cu3V2O8
|
Li-Si |
LiPO3, LiSO4, Li2CO3, LiF |
Thin film/ 80 |
990 |
500 |
2.0 |
2.971 |
2.4 |
- |
[30] |
|
18 |
ZrS3
|
Li-Si |
LiCl-KCl |
Pellet/ - |
1285.2 |
500 |
1.0 |
1.8 |
1.7 |
700 |
[31] |
|
19 |
CoNi2S4
|
Li-Si |
LiCl-KCl |
Pellet/ - |
1144.8 |
500 |
1.25 |
2.58 |
- |
500 |
[32] |
REFERENCES
1. RA. Guidotti, and P. Masset, “Thermally Activated (“Thermal”) Battery Technology Part I: An Overview,” J Power Sources, 161 [2] 1443-49 (2006).  2. P. Masset, and RA. Guidotti, “Thermally Activated (“Thermal”) Battery Technology Part II: Molten Salt Electrolytes,” J Power Sources, 164 [1] 397-414 (2007).  3. PJ. Masset, and RA. Guidotti, “Thermally Activated (“Thermal”) Battery Technology Part IIIa: FeS2 Cathode Material,” J Power Sources, 177 [2] 595-609 (2008).  4. PJ. Masset, and RA. Guidotti, “Thermally Activated (“Thermal”) Battery Technology Part IIIb: Sulfur and Oxide-based Cathode Materials,” J Power Sources, 178 [1] 456-66 (2008).  5. RA. Guidotti, and PJ. Masset, “Thermally Activated (“Thermal”) Battery Technology Part IV: Anode Materials,” J Power Sources, 183 [1] 388-98 (2008).  6. SS. Wang, and RN. Seefurth, “Electrochemical Studies of FeS2 Electrodes in Various Sulfide-Containing Molten Salts,” J Electrochem Soc, 134 [3] 530-35 (1987).  7. SH. Chae, SH. Kang, HW. Cheong, YS. Han, and DH. Yoon, “Thermal Batteries with Ceramic Felt Separators - Part 1: Wetting, Loading Behavior and Chemical Stability,” Ceram Int, 43 [5] 4015-22 (2017).  8. SH. Kang, SH. Chae, HW. Cheong, KH. Kim, YS. Han, SM. Lee, DH. Yoon, and J. Yi, “Thermal Batteries with Ceramic Felt Separators - Part 2: Ionic Conductivity, Electrochemical and Mechanical Properties,” Ceram Int, 43 [5] 4023-28 (2017).  9. YS. Choi, HR. Yu, and HW. Cheong, “Electrochemical Properties of a Lithium-Impregnated Metal Foam Anode for Thermal Batteries,” J Power Sources, 276 102-4 (2015).  10. Y. Choi, S. Cho, and YS. Lee, “Effect of the Addition of Carbon Black and Carbon Nanotube to FeS2 Cathode on the Electrochemical Performance of Thermal Battery,” J Ind Eng Chem, 20 [5] 3584-89 (2014).  11. J. Ko, IY. Kim, HM. Jung, H. Cheong, and YS. Yoon, “Thin Cathode for Thermal Batteries Using a Tape Casting Process,” Ceram Int, 43 [7] 5789-93 (2017).  12. I. Oh, J. Cho, K. Kim, J. Ko, H. Cheong, YS. Yoon, and HM. Jung, “Poly(Imide-co-Siloxane) as a Thermo-Stable Binder for Thin Layer Cathode of Thermal Batteries,” Energies, 11 [11] 3154(2018).  13. J. Ko, IY. Kim, H. Cheong, and YS. Yoon, “Organic Binder-free Cathode Using FeS2-MWCNTs Composite for Thermal Batteries,” J Am Ceram Soc, 100 [10] 4435-41 (2017).  14. T. Yang, L. Cai, and RE. White, “Mathematical Modeling of the LiAl/FeS2 High Temperature Battery System,” J Power Sources, 201 322-31 (2012).  15. EL. Reinholz, SA. Roberts, CA. Apblett, JB. Lechman, and PR. Schunk, “Composition and Manufacturing Effects on Electrical Conductivity of Li/FeS2 Thermal Battery Cathode,” J Electrochem Soc, 163 [8] A1723-29 (2016).  16. J. Hu, Y. Chu, Q. Tian, J. Wang, Y. Li, Q. Wu, L. Zhao, and Y. Zhu, “Film Cathode for Thermal Batteries Using a Screen-Printing Process,” Mater Lett, 215 296-99 (2018).  17. S. Xie, Y. Deng, J. Mei, Z. Yang, WM. Lau, and H. Liu, “Facile Synthesis of CoS2/CNTs Composite and its Exploitation in Thermal Battery Fabrication,” Composites, Part B, 93 203-9 (2016).  18. S. Xie, Y. Deng, J. Mei, Z. Yang, WM. Lau, and H. Liu, “Carbon Coated CoS2 Thermal Battery Electrode Material with Enhanced Discharge Performances and Air Stability,” Electrochim Acta, 231 287-93 (2017).  19. T. Yu, Z. Yu, Y. Cao, H. Liu, X. Liu, Y. Cui, C. Wang, and Y. Cui, “Electrochemical Performances and Air Stability of Fe-deped CoS2 Cathode Materials for Thermal Batteries,” Int J Electrochem Sci, 13 7590-97 (2018).  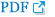 20. J. Hu, L. Zhao, Y. Chu, Q. Tian, J. Wang, Y. Li, Q. Wu, and Y. Zhu, “Preparation and Electrochemical Properties of a New Fe0.5Co0.5S2 Cathode Material for Thermal Batteries,” J Alloys Compd, 762 109-114 (2018).  21. Y. Xie, Z. Liu, H. Ning, H. Huang, and L. Chen, “Suppressing Self-Discharge of Li-B/CoS2 Thermal Batteries by Using a Carbon-Coated CoS2 Cathode,” RSC Adv, 8 [13] 7173-78 (2018).  22. C. Jin, L. Zhou, L. Fu, J. Zhu, D. Li, and W. Yang, “The Acceleration Intermediate Phase (NiS and Ni3S2) Evolution by Nanocrystallization in Li/NiS2 Thermal Batteries with High Specific Capacity,” J Power Sources, 352 83-9 (2017).  23. C. Jin, L. Fu, J. Zhu, W. Yang, D. Li, and L. Zhou, “A Hierarchical Carbon Modified Nano-NiS2 Cathode with High Thermal Stability for a High Energy Thermal Battery,” J Mater Chem A, 6 [16] 7123-32 (2018).  24. JL. Payne, JD. Percival, K. Giagloglou, CJ. Crouch, GM. Carins, RI. Smith, R. Comrie, RKB. Gover, and JTS. Irvine, “In-situ Thermal Battery Discharge Using NiS2 as a Cathode Material,” ChemElectroChem, 4 [8] 1-9 (2017).  25. C. Jin, L. Zhou, L. Fu, J. Zhu, and D. Li, “Synthesis and Discharge Performances of NiCl2 by Surface Modification of Carbon Coating as Cathode Material of Thermal Battery,” Appl Surf Sci, 402 308-13 (2017).  26. J. Hu, Y. Chu, Q. Tian, S. Guo, M. Yang, X. Wang, L. Zhao, and Y. Zhu, “Electrochemical Properties of the NiCl2 Cathode with Nickel Foam Substrate for Thermal Batteries,” Mater Lett, 207 198-201 (2017).  27. W. Liu, H. Liu, S. Bi, L. Cao, and Y. Sun, “Variable-Temperature Preparation and Performance of NiCl2 as a Cathode Material for Thermal Batteries,” Sci China Mater, 60 [3] 251-57 (2017).  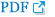 28. K. Giagloglou, JL. Payne, C. Crouch, RKB. Gover, PA. Connor, and JTS. Irvine, “Transition Metal Chlorides NiCl2, KNiCl3, Li6VCl8 and Li2MnCl4 as Alternative Cathode Materials in Primary Li Thermal Batteries,” J Electrochem Soc, 165 [14] A3510-16 (2018).  29. T. Hillel, and Y. Ein-Eli, “Copper Vanadate as Promising High Voltage Cathodes for Li Thermal Batteries,” J Power Sources, 229 112-16 (2013).  30. J. Dai, M. Lai, R. LaFollette, and D. Reisner, “Thin Film Copper Vanadium Oxide Electrodes for Thermal Batteries,” ECS Trans, 33 [27] 3-9 (2011).  31. K. Giagloglou, JL. Payne, C. Crouch, RKB. Gover, PA. Connor, and JTS. Irvine, “Zirconium Trisulfide as a Promising Cathode Material for Li Primary Thermal Batteries,” J Electrochem Soc, 163 [14] A3126-30 (2016).  32. K. Giagloglou, JL. Payne, C. Crouch, RKB. Gover, PA. Connor, and JTS. Irvine, “Synthesis and Electrochemical Study of CoNi2S4 as a Novel Cathode Material in a Primary Li Thermal Battery,” J Electrochem Soc, 164 [9] A2159-63 (2017).  33. X. Zheng, Y. Zhu, Y. Sun, and Q. Jiao, “Hydrothermal Synthesis of MoS2 with Different Morphology and its Performance in Thermal Battery,” J Power Sources, 395 318-27 (2018).  34. K. Hasegawa, and S. Noda, “Lithium Ion Batteries Made of Electrodes with 99 wt% Active Materials and 1 wt% Carbon Nanotubes without Binder or Metal Foils,” J Power Sources, 321 155-62 (2016).  35. P. Sehrawat, C. Julien, and SS. Islam, “Carbon Nanotubes in Li-Ion Batteries: A Review,” Mater Sci Eng, B, 213 12-40 (2016).  36. IY. Kim, SY. Shin, JH. Ko, KS. Lee, SP. Woo, DK. Kim, and YS. Yoon, “Functional Li-M (Ti, Al, Co, Ni, Mn, Fe)-O Energy Materials,” J Korean Ceram Soc, 54 [1] 9-22 (2017).  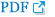 37. KS. Lee, SY. Shin, and YS. Yoon, “Fe3O4 Nanoparticles on MWCNTs Backbone for Lithium Ion Batteries,” J Korean Ceram Soc, 53 [3] 376-80 (2016).   38. JH. Kim, and DK. Kim, “Conversion-Alloying Anode Materials for Na-Ion Batteries: Recent Progress, Challenges, and Perspective for the Future,” J Korean Ceram Soc, 55 [4] 307-24 (2018).   39. T. Sadhasivam, MJ. Park, JY. Shin, JE. Jin, SC. Kim, MD. Kurkuri, SH. Roh, and HY. Jung, “High Charge Acceptance though Interface Reaction on Carbon Coated Negative Electrode for Advanced Lead-Carbon Battery System,” Electrochim Acta, 295 367-75 (2019).  40. J. Yin, N. Lin, W. Zhang, Z. Lin, Z. Zhang, Y. Wang, J. Shi, J. Bao, and H. Lin, “Highly Reversible Lead-Carbon Battery Anode with Lead Grafting on the Carbon Surface,” J Energy Chem, 27 1674-83 (2018).  41. W. Wang, Y. Liang, Y. Kang, L. Liu, Z. Xu, X. Tian, W. Mai, H. Fu, H. Lv, K. Teng, X. Jiao, and F. Li, “Carbon-Coated SnO2@Carbon Nanofibers Produced by Electrospinning-Electrospraying Method for Anode Materials of Lithium-Ion Batteries,” Mater Chem Phys, 223 762-70 (2019).  42. H. Akbulut, D. Nalci, A. Guler, S. Duman, and MO. Guler, “Carbon-Silicon Composite Anode Electrodes Modified with MWCNT for High Energy Battery Applications,” Appl Surf Sci, 446 222-29 (2018).  43. YN. Lee, SP. Woo, YS. Yoon, and SH. Kim, “Significant Improvement in Reversibility of MWCNTs-Sn Compound Composite Electrode: Nanostructure Effect MWCNT-Sn Compound Composite on High Initial Reversible Capacity,” J Alloys Compd, 777 1098-107 (2019).  44. X. Yan, Y. Wang, T. Yu, H. Chen, Z. Zhao, and S. Guan, “Polyimide Binder by Combining with Polyimide Separator for Enhancing the Electrochemical Performance of Lithium Ion Batteries,” Electrochim Acta, 216 1-7 (2016).  45. J. Choi, MH. Ryou, B. Son, J. Song, JK. Park, KY. Cho, and YM. Lee, “Improved High-Temperature Performance of Lithium-Ion Batteries through Use of a Thermally Stable Co-Polyimide-based Cathode Binder,” J Power Sources, 252 138-43 (2014).  46. J. Liu, Q. Zhang, and YK. Sun, “Recent Progress of Advanced Binders for Li-S Batteries,” J Power Sources, 396 19-32 (2018).  47. M. Zheng, Y. Wang, J. Reeve, H. Souzandeh, and WH. Zhong, “A Polymer-Alloy Binder for Structures-Properties Control of Battery Electrode,” Energy Storage Mater, 14 149-58 (2018).  48. G. Hernandez, N. Lago, D. Shanmukaraj, M. Armand, and D. Mecerreyes, “Polyimide-Polyether Binder-Diminishing the Carbon Content in Lithium-Sulfur Batteries,” Mater Today Energy, 6 264-70 (2017).  49. J. Luis, G. Urbano, JL. Gomez-Camer, C. Botas, and T. Rojo, “Graphene Oxide-Carbon Nanotubes Aerogels with High Sulfur Loadings Suitable as Binder-free Cathodes for High Performance Lithium-Sulfur Batteries,” J Power Sources, 412 408-15 (2019).  50. L. Guo, H. Sun, C. Qin, W. Li, F. Wang, W. Song, J. Du, F. Zhong, and Y. Ding, “Flexible Fe3O4 Nanoparticles/N-doped Carbon Nanofibers Hybrid Film as Binder-free Anode Materials for Lithium-Ion Batteries,” Appl Surf Sci, 459 263-70 (2018).  51. Y. Liu, X. Chi, Q. Han, Y. Du, J. Yang, and Y. Liu, “Vertically Self-Standing C@NiCo2O4 Nanoneedle Arrays as Effective Binder-free Cathode for Rechargeable Na-O2 Batteries,” J Alloys Compd, 772 693-702 (2019).  52. J. Nong, P. Xie, AS. Zhu, MZ. Rong, and MQ. Zhang, “Highly Conductive Doped Carbon Framework as Binder-free Cathode for Hybrid Li-O2 Battery,” Carbon, 142 177-89 (2019).  53. B. Li, Q. Xiao, and Y. Luo, “A Modified Synthesis Process of Three-Dimensional Sulfur/Graphene Aerogel as Binder-free Cathode for Lithium Sulfur Batteries,” Mater Des, 153 9-14 (2018).  54. TG. Kim, E. Samuel, B. Joshi, CW. Park, MW. Kim, MT. Swihart, WY. Yoon, and SS. Yoon, “Supersonically Sprayed rGO-Zn2Sn4 Composites as Flexible, Binder-free, Scalable, and High-Capacity Lithium Ion Battery Anodes,” J Alloys Compd, 766 331-40 (2018).  55. CC. Li, and YW. Wang, “Binder Distributions in Water-based and Organic-based LiCoO2 Electrode Sheets and their Effects on Cell Performance,” J Electrochem Soc, 158 A1361-70 (2011).  56. M. Muller, L. Pfaffmann, S. Jaiser, M. Baunach, V. Trouillet, F. Scheiba, P. Scharfer, W. Schabel, and W. Bauer, “Investigation of Binder Distribution in Graphite Anodes for Lithium-Ion Batteries,” J Power Sources, 340 1-5 (2017).  57. B. Lestriez, “Functions of Polymers in Composite Electrodes of Lithium Ion Batteries,” C R Chim, 13 [11] 1341-50 (2010).  58. SL. Chou, Y. Pan, JZ. Wang, HK. Liu, and SX. Dou, “Small Things Mask a Big Difference: Binder Effects on the Performance of Li and Na Batteries,” Phys Chem Chem Phys, 16 [38] 20347-59 (2014).  59. Z. Zhang, T. Zeng, Y. Lai, M. Jia, and J. Li, “A Comparative Study of Different Binders and Their Effects on Electrochemical Properties of LiMn2O4 Cathode in Lithium Ion Batteries,” J Power Sources, 247 1-8 (2014).  60. G. Liu, H. Zheng, X. Song, and VS. Battaglia, “Particles and Polymer Binder Interaction: A Controlling Factor in Lithium-Ion Electrode Performance,” J Electrochem Soc, 159 [3] A214-21 (2012).  61. SH. Lee, C. Huang, C. Johnston, and PS. Grant, “Spray Printing and Optimization of Anodes and Cathodes for High Performance Li-Ion Batteries,” Electrochim Acta, 292 546-57 (2018).  62. AF. Leonard, and N. Job, “Safe and Green Li-Ion Batteries based on LiFePO4 and Li4Ti5O12 Sprayed as Aqueous Slurries with Xanthan Gum as Common Binder,” Mater Today Energy, 12 168-78 (2019).  63. SD. Kim, JG. Lee, TG. Kim, K. Rana, JY. Jeong, JH. Park, SS. Yoon, and JH. Ahn, “Additive-free Electrode Fabrication with Reduced Graphene Oxide Using Supersonic Kinetic Spray for Flexible Lithium-Ion Batteries,” Carbon, 139 195-204 (2018).  64. B. Joshi, E. Samuel, TG. Kim, CW. Park, YI. Kim, MT. Swihart, WY. Yoon, and SS. Yoon, “Supersonically Spray-Coated Zinc Ferrite/Graphitic-Carbon Nitride Composite as a Stable High-Capacity Anode Material for Lithium-Ion Batteries,” J Alloys Compd, 768 525-34 (2018).  65. H. Shi, S. Niu, W. Lv, G. Zhou, C. Zhang, Z. Sun, F. Li, F. Kang, and QH. Yang, “Easy Fabrication of Flexible and Multilayer Nanocarbon-based Cathodes with a High Unreal Sulfur Loading by Electrostatic Spraying for Lithium-Sulfur Batteries,” Carbon, 138 18-25 (2018).  66. CY. Jung, TS. Zhao, L. An, L. Zeng, and ZH. Wei, “Screen Printed Cathode for Non-Aqueous Lithium-Oxygen Batteries,” J Power Sources, 297 174-80 (2015).  67. RE. Sousa, J. Oliveira, A. Goren, D. Miranda, MM. Silva, L. Hilliou, CM. Costa, and S. Lanceros-Mendez, “High Performance Screen Printable Lithium-Ion Battery Cathode Ink based on C-LiFePO4
,” Electrochim Acta, 196 92-100 (2016).  68. A. Goren, J. Mendes, HM. Rodrigues, RE. Sousa, J. Oliveira, L. Hilliou, CM. Costa, MM. Silva, and S. Lanceros-Mendez, “High Performance Screen-Printed Electrodes Prepared by a Green Solvent Approach for Lithium-Ion Batteries,” J Power Sources, 334 65-77 (2016).  69. KY. Kang, YG. Lee, DO. Shin, JC. Kim, and KM. Kim, “Performance Improvements of Pouch-Type Flexible Thin-Film Lithium-Ion Batteries by Modifying Sequential Screen Printing Process,” Electrochim Acta, 138 294-301 (2014).  70. Z. Tehrani, T. Korochkina, S. Govindarajan, DJ. Thomas, J. O’Mahony, J. Kettle, TC. Claypole, and DT. Gethin, “Ultra-Thin Flexible Screen Printed Rechargeable Polymer Battery for Wearable Electronic Applications,” Org Electron, 26 386-94 (2015).  71. B. Bitsch, J. Dittmann, M. Schmitt, P. Scharfer, W. Schabel, and N. Willenbacher, “A Novel Slurry Concept for the Fabrication of Lithium-Ion Battery Electrodes with Beneficial Properties,” J Power Sources, 265 81-90 (2014).  72. A. Ponrouch, and MR. Palacin, “On the Impact of the Slurry Mixing Procedure in the Electrochemical Performance of Composite Electrodes for Li-Ion Batteries: A Case Study for Mesocarbon Microbeads (MCMB) Graphite and Co3O4
,” J Power Sources, 196 9682-88 (2011).  73. KY. Cho, YI. Kwon, JR. Youn, and YS. Song, “Evaluation of Slurry Characteristics for Rechargeable Lithium-Ion Batteries,” Mater Res Bull, 48 [8] 2922-26 (2013).  74. W. Bauer, and D. Notzel, “Rheological Properties and Stability of NMP Based Cathode Slurries for Lithium Ion Batteries,” Ceram Int, 40 [3] 4591-98 (2014).  75. K. Okubo, H. Wang, K. Hayashi, M. Inada, N. Enomoto, G. Hasegawa, T. Osawa, and H. Takamura, “A Dense NASICON Sheet Prepared by Tape-Casting and Low Temperature Sintering,” Electrochim Acta, 278 176-81 (2018).  76. A. Rincon, R. Moreno, ASA. Chinelatto, CF. Gutierrez, E. Rayon, MD. Salvador, and A. Borrell, “Al2O3-3YTZP-Graphene Multilayers Produced by Tape Casting and Spark Plasma Sintering,” J Eur Ceram Soc, 34 [10] 2427-34 (2014).  77. MR. Somalu, A. Muchtar, WRW. Daud, and NP. Brandon, “Screen-Printing Inks for the Fabrication of Solid Oxide Fuel Cell Films: A Review,” Renewable Sustainable Energy Rev, 75 426-39 (2017).  78. W. Wang, S. Chen, J. Li, and W. Wang, “Fabrication of Catalyst Coated Membrane with Screen Printing Method in a Proton Exchange Membrane Fuel Cell,” Int J Hydrogen Energy, 40 [13] 4649-58 (2015).  79. EF. Mine, Y. Ito, Y. Teranishi, M. Sato, and T. Shimizu, “Surface Coating and Texturing on Stainless-Steel Plates to Decrease the Contact Resistance by Using Screen Printing,” Int J Hydrogen Energy, 42 [31] 20224-29 (2017).  80. DH. Lee, JS. Choi, H. Chae, CH. Chung, and SM. Cho, “Highly Efficient Phosphorescent Polymer OLEDs Fabricated by Screen Printing,” Displays, 29 [5] 436-39 (2008).  81. S. Ohta, S. Komagata, J. Seki, T. Saeki, S. Morishita, and T. Asaoka, “All-Solid-State Lithium Ion Battery Using Garnet-Type Oxide and Li3BO3 Solid Electrolytes Fabricated by Screen-Printing,” J Power Sources, 238 53-6 (2013).  82. T. Syrovy, T. Kazda, L. Syrova, J. Vondrak, L. Kubac, and M. Sedlarikova, “Cathode Material for Lithium Ion Accumulators Prepared by Screen Printing for Smart Textile Applications,” J Power Sources, 309 192-201 (2016).  83. MH. Sayed, EVG. Robert, PJ. Dale, and L. Gutay, “Cu2SnS3 Based Thin Film Solar Cells from Chemical Spray Pyrolysis,” Thin Solid Films, 669 436-39 (2019).  84. Z. Liang, Z. Bi, K. Gao, Y. Fu, P. Guan, X. Feng, Z. Chai, G. Xu, and X. Xu, “Interface Modification via Al2O3 with Retarded Charge Recombination for Mesoscopic Perovskite Solar Cells Fabricated with Spray Deposition Process in the Air,” Appl Surf Sci, 463 939-46 (2019).  85. KY. Bae, MW. Kim, BH. Kim, SH. Cho, SS. Yoon, and WY. Yoon, “Effect of Electrostatic Spray Deposited Nafion Coating on Non-Lithiated LiV3O8 Cathode in Lithium- Metal Rechargeable Batteries,” Solid State Ionics, 331 66-73 (2019).  86. X. Wu, F. Li, W. Wu, and T. Guo, “Flexible Organic Light Emitting Diodes Based on Double-Layered Graphene/PEDOT:PSS Conductive Film Formed by Spray-Coating,” Vacuum, 101 53-6 (2014).  87. A. Falco, AM. Zaidi, P. Lugli, and A. Abdellah, “Spray Deposition of Polyethylenimine Thin Films for the Fabrication of Fully-Sprayed Organic Photodiodes,” Org Electron, 23 186-92 (2015).  88. T. Bayer, R. Selyanchyn, S. Fujikawa, K. Sasaki, and SM. Lyth, “Spray-Painted Graphene Oxide Membrane Fuel Cells,” J Membr Sci, 541 347-57 (2017).  89. AB. Tahar, A. Romdhane, N. Lalaoui, N. Reverdy-Bruas, AL. Goff, M. Holzinger, S. Cosnier, D. Chaussy, and N. Belgacem, “Carbon Nanotube-based Flexible Biocathode for Enzymatic Biofuel Cells by Spray Coating,” J Power Sources, 408 1-6 (2018).  90. Q. Guo, P. Guo, J. Li, H. Yin, J. Liu, F. Xial, D. Shen, and N. Li, “Fe3O4-CNTs Nanocomposites: Inorganic Dispersant Assisted Hydrothermal Synthesis and Application in Lithium Ion Batteries,” J Solid State Chem, 213 104-9 (2014).  91. Q. Liu, Q. Jiang, L. Jiang, J. Peng, Y. Gao, Z. Duan, and X. Lu, “Preparation of SnO2@rGO/CNTs/S Composite and Application for Lithium-Sulfur Battery Cathode Material,” Appl Surf Sci, 462 393-98 (2018).  92. M. Kazazi, ZA. Zafar, M. Delshad, J. Cervenka, and C. Chen, “TiO2/CNT Nanocomposite as an Improved Anode Material for Aqueous Rechargeable Aluminum Batteries,” Solid State Ionics, 320 64-9 (2018).  93. MY. Son, JH. Choi, and YC. Kang, “Electrochemical Properties of Bare Nickel Sulfide and Nickel Sulfide-Carbon Composite Prepared by One-Pot Spray Pyrolysis as Anode Materials for Lithium Secondary Batteries,” J Power Sources, 251 480-87 (2014).  94. Y. Yamaguchi, T. Takeuchi, H. Sakaebe, H. Kageyama, H. Senoh, T. Sakai, and K. Tatsumi, “Ab Initio Simulations of Li/Pyrite-MS2 (M=Fe, Ni) Battery Cells,” J Electrochem Soc, 157 [6] A630-35 (2010).  95. AA. AbdelHamid, X. Yang, J. Yang, X. Chen, and JY. Ying, “Graphene-Wrapped Nickel Sulfide Nanoprisms with Improved Performance for Li-Ion Battery Anodes and Supercapacitors,” Nano Energy, 26 425-37 (2016).  96. T. Takeuchi, H. Sakaebe, H. Kageyama, T. Sakai, and K. Tatsumi, “Preparation of NiS2 Using Spark-Plasma-Sintering Process and its Electrochemical Properties,” J Electrochem Soc, 155 [9] A679-84 (2008).  97. SW. Oh, ST. Myung, SM. Oh, KH. Oh, K. Amine, B. Scrosati, and YK. Sun, “Double Carbon Coating of LiFe-PO4 as High Rate Electrode for Rechargeable Lithium Batteries,” Adv Mater, 22 [43] 4842-45 (2010).  98. X. Zhang, X. Zhang, XG. Wang, Z. Xie, and Z. Zhou, “NiFe2O4-CNT Composite: An Efficient Electrocatalyst for Oxygen Evolution Reactions in Li-O2 Batteries Guided by Computations,” J Mater Chem A, 4 [24] 9390-93 (2016).  99. J. Yang, Y. Ouyang, H. Zhang, H. Xu, Y. Zhang, and Y. Wang, “Novel Fe2P/Graphitized Carbon Yolk/Shell Octahedral for High-Efficiency Hydrogen Production and Lithium Storage,” J Mater Chem A, 4 [25] 9923-30 (2016).  100. Z. Liu, T. Lu, T. Song, XY. Yu, XW. Lou, and U. Paik, “Structure-Designed Synthesis of FeS2@C Yolk-Shell Nanoboxes as a High-Performance Anode for Sodium-Ion Batteries,” Energy Environ Sci, 10 [7] 1576-80 (2017).  101. Y. Li, YS. Hu, MM. Titirici, L. Chen, and X. Huang, “Hard Carbon Microtubes Made from Renewable Cotton as High-Performance Anode Material for Sodium-Ion Batteries,” Adv Energy Mater, 6 [18] 1600659(2016).  102. J. Shan, Y. Liu, Y. Su, P. Liu, X. Zhuang, D. Wu, F. Zhang, and X. Feng, “Graphene-Directed Two-Dimensional Porous Carbon Frameworks for High-Performance Lithium-Sulfur Battery Cathodes,” J Mater Chem A, 4 [1] 314-20 (2016).  103. Y. Sakurai, H. Ohtsuka, and J. Yamaki, “Rechargeable Copper Vanadate Cathode for Lithium Cell,” J Electrochem Soc, 135 [1] 32-6 (1988).  104. M. Eguchi, A. Komamura, T. Miuru, and T. Kishi, “Lithiation Characteristics of Cu5V2O10
,” J Electrochim Acta, 41 [6] 857-61 (1996).  105. F. Gao, J. Shi, H. Liu, S. Qiang, L. Gao, S. Bi, and W. Liu, “A Novel and Safety Lithium Thermal Battery Electrolyte - Li7La3Zr2O12 Prepared by Solid State Method,” Solid State Ionics, 326 131-35 (2018). 
|
|






























