1. Introduction
Heat insulating materials of high-temperature type have been used as generally fiber-based or fumed silica-based porous compacted insulation frequently for power generation plants, oil refinery plants, and steel mills. The fumed silica based porous compact insulation is fabricated by dry pressing of powder mixture. The insulation or mineral wool is produced in a board type or in a quilt type with the outer skin layer surrounded by glass fiber fabric, which contain nano-sized fumed silica powders capable of greatly reducing heat flows by conduction, convection and radiation as the main composition, adding opacifiers shutting off infrared radiation heat, and reinforcing with glass fibers and binders to increase green compact strength. Fumed silica particles as a type of silica powders form a structure with a few ten or a few hundred primary particles of 7 ~ 40 nm in size loosely agglomerated like a tortuous path to lower thermal conductivities by elongating the pathway for heat conduction and suppressing heat transfer by convection of heat due to having pore sizes of a nano level.1-3) Fumed silica at room temperature is known to have low thermal conductivity values of 0.01 ~ 0.03 W/m·K. Many studies have been performed for improvement of heat insulation by adding opacifiers of high refractive index such as TiO2, SiC, ZrO2, C, etc. capable of reducing radiative heat transfer by scattering or absorbing radiation.3-11) The infrared ray irradiated onto opacifiers have difficulty in passing through the heat insulation plate due to scattering and reflection or absorption, suppressing radiative heat transfer. Therefore, the radiated energy is affected by contents of opacifiers for heat insulation material and plate thickness according to the Beer-Lambert law stating that absorbance of light is proportional to the concentration and thickness of the medium through which light is passing. As application temperature is raised, the radiation spectrum intensity is increased by 4 orders of magnitude and the wavelengths of radiated rays are shortened so that infrared extinction of the opacifier is an important element for the heat insulating property. Since the radiated rays have a continuous spectrum rather than a characteristic spectrum, it is not easy to determine a specific particle size having the greatest infrared extinction at a application temperature. The particle size has also a distribution. Zhao et al. have studied the effects of infrared wavelength or measurement temperature on extinction coefficient by adding TiO2, SiC, ZrO2 opacifier powders of various particle sizes and particle forms to silica aerogel.5) In their study, the result for SiC is presented where the infrared extinction coefficient as a function of measurement temperature is the largest for 4 μm at 400°C, and for 3 μm at 600°C. As green compacts should have an appropriate strength to use materials in powder form such as fumed silica as a high-temperature heat insulation material, 2 ~ 20 wt% of glass fiber or silica fiber is added as reinforcement.2,7) To prevent desorption of fumed silica particles and to reinforce strength, inorganic binders such as water glass (alkali silicate), phosphates, borates, etc. are being used.12,13) To grant initial green strength, organic binders such as PVA, cellulose, etc. may also be used. However, the use of the latter is limited to 200°C due to thermal stability problems. While mechanical characteristics are improved by addition of fibers and inorganic binders, negative effects on light weight and heat insulation property of the fumed silica may be resulted.14) Such compressed type of heat insulation is produced and sold globally by Microtherm of UK, Porextherm of Germany, and domestically by Morgan, Kyungdongone, KCC etc. Products with low thermal conductivities of 0.02 ~ 0.04 W/m·K for the application in the temperature range of 200 ~ 800°C and with different mechanical strengths are being produced.
In the present study, to investigate heat insulation property of fumed silica based insulation, various compacted plates were prepared by pressing the dried mixtures of chopped glass fiber, fumed silica, and SiC powder using a square steel mold of 300 × 300 mm, followed by evaluation of thermal conductivities at 400 and 600°C by guarded hot plate method. The compacts of mixed powders in 5 compositions for the surface temperature measurement were formed using a square steel mold of 100 × 100 mm. The surface temperature measurement of the plates was performed by exposing the plate bottom to the set temperature of an electric furnace, 400 and 600°C. By checking for the correlation coefficients between thermal conductivity and surface temperature of heat insulation plates by using the independent surface temperature measuring instrument, the evaluation of heat insulation property was possible for the plates of various compositions. By measuring surface temperatures of the heat insulation materials with addition of SiC powders in the range of 1.3 ~ 17.5 μm having 7 types of different sizes, the effects of SiC particles on heat insulation property were discussed based on the correlation coefficients between thermal conductivity and surface temperature. In addition, heat insulation plates were prepared by using inorganic binders including 3 types of alkali silicates and 2 types of phosphates, and surface temperatures were measured followed by evaluation of the effects of binders on flexural strength and heat insulation property.
2. Experimental Procedure
2.1. Raw material characterization
As the raw materials for preparation of heat insulation materials, fumed silica (Tokuyama), SiC (Sinxing advanced material), and glass fiber of 12 μm in diameter (Owens Corning) were used. The microstructure of the raw materials was investigated by SEM (SEM300, Topcon) as shown in Fig. 1. Particle size distributions of SiC powders were measured by PSA (Particle Size Analysis, LA-950, Horiba). Specific surface areas of SiC powders were measured by using BET (Brunauer Emmett and Teller, TriStar3000, MICRO-MERITICS) after pretreatment at 120°C for 24 h. The fumed silica has a specific surface area of 312 m2/g and a primary particle size of ~ 20 nm with an agglomerated-type structure in 3D chain form. For SiC powders, 7 different sizes including 1.3, 2.2, 3.9, 5.1, 8.2, 13.1, and 17.5 μm were used. To improve green strengths of the specimens, 3 types of alkali silicates including potassium silicate (PS) (27 ~ 29%, Junsei chemical), sodium silicate (S.S-1) (R-300, Youngil chemical), and sodium silicate (S.S-2) (ECO-300, Youngil chemical) as well as 2 types of phosphates including aluminum phosphate (A.P-1) (542, Aremco) and phosphate compounds (A.P-2) (503-VFG-C, Aremco)were used as inorganic binders, which are shown in Table 1.
2.2. Preparation of Mixtures
To study thermal conductivity of heat insulation plates as a function of compositions, design of experiment (DOE) was applied to mixing of the raw materials consisting of fumed silica, glass fiber and SiC powders of 4.4 μm in size. 10 compositions of heat insulation plates in the range of 80 ~ 96 wt% for fumed silica, 2 ~ 18 wt% for glass fiber, and 2 ~ 18 wt% for SiC were prepared and their thermal conductivities were measured. 5 representative compositions with distinctive thermal conductivity values are selected from 10 compositions for measurement of surface temperature as shown in Table 2. For binder mixing, 5 wt% and 10 wt% of binders were added and mixed for the mixed powder having 80% of fumed silica, 10% of glass fiber and 10% of SiC (composition for Sample 2).
2.3. Plate preparation and characterization
After mixing the raw materials at a speed higher than 2000 rpm in a dry mixer of 1 m3 capacity and placing the powder in a square mold of 300 × 300 mm, square plates of ~ 25 mm thickness were prepared by compression at 20 atm using a biaxial press. To remove water contained in the plates, they were subjected to drying at 120°C for more than 24 h. After cutting the heat insulation plates into a size of 250 × 250 mm, thermal conductivity was measured at temperatures of 400°C and 600°C, respectively, using the Guarded Hot Plate method (GHP 456 Titan, Germany). For evaluation of mechanical strengths for the prepared heat insulation plates, measurements were made by using a universal tester (R&B Co., Ltd., RB 301) after the heat insulation plates were cut into a size of 3 cm × 9 cm × 0.6 (± 0.3 cm).
2.4. Surface temperature measurements
To prepare the specimens for surface temperature measurement, 20 g of raw powders was placed in a 1 L mixing pulverizer and mixed at about 1000 rpm for 5 min. To find a difference in mixedness between the powder mixed at 2000 rpm in a dry mixer of 1 m3 capacity and the powder mixed at 1000 rpm in a 1 L mixing pulverizer, heat insulation plates were prepared per formulation and surface temperatures were compared at 400°C and 600°C. Here, no large problem is considered to exist in the mixedness as the surface temperatures of heat insulation plates differed by less than 1%. Square plates were prepared by placing the mixed powder in a square mold of 110 × 110 mm followed by compression molding using a press so that the thickness of heat insulation plate became 0.6 ± 0.3 cm. To remove water contained in the specimens, drying was conducted at 120°C for more than 4 h. Surface temperature of heat insulation materials was measured by fixing a thermographic camera on the electric furnace connected to a controller. The relevant temperature for electric furnace was set at a temperature rise rate of 20°C/min for inside temperatures and of 4°C/min for outside temperatures by using the controller, and was held for 4 h to stabilize temperatures of the electric furnace. After stabilization, the heat insulation material specimens were placed on an aluminum substrate to be stabilized for 20 minutes, followed by measurement of surface temperatures for the heat insulation material. Schematic diagram for the employed apparatus to measure surface temperatures is shown in Fig. 2. The surface temperature is a value of average temperature measured for the area of 70 × 70 mm in the heat insulation plate for 1 minute using a thermographic camera.
3. Results and Discussion
3.1. Thermal conductivity vs. surface temperature
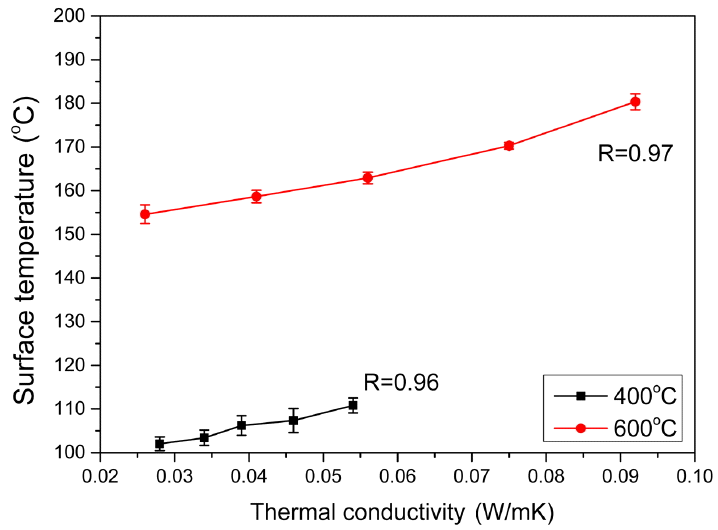
Since the specimens for measurement of high temperature thermal conductivity have a large size of 250 × 250 × 25 mm taking 1 ~ 2 days for measuring one specimen, a long time is required for measuring several specimens. Surface temperature measurement method drastically reduces the specimen size and the measurement time for evaluation of heat insulation properties. In Table 2, measured values of thermal conductivity and the corresponding surface temperature values of the specimens at 400°C and 600°C. Here, the values represent an average of 4-time measurements of surface temperature for 2 types of specimens per compositions by using a thermographic camera, with the specimen thickness being 6 ± 0.3 mm. At 400°C, the surface temperatures of the specimens were measured to be 102 ~ 110°C, while the surface temperatures of 150 ~ 180°C were measured at 600°C. As the temperature was increased, differences in surface temperatures per compositions were shown to be large. Shown in Fig. 3 are the average and the standard deviation for 4-time measurements of surface temperature at 400°C and 600°C using a thermographic camera. Correlation coefficients between thermal conductivity and surface temperature at 400°C and 600°C are high as 0.96 and 0.97, respectively. Thus, it was confirmed that surface temperature measurements of the specimen could be an easy and convenient means for evaluation of heat insulation property of the specimens.
According to thermal conductivity results as a function of compositions, low thermal conductivities were obtained for the compositions where SiC content was high, ceramic fiber content was low and fumed silica content was increased. For heat transfer at high temperature of 600°C the radiative heat transfer was more dominant than conductivity heat transfer, suggesting that SiC plays an important role in heat transfer.
3.2. Density vs. surface temperature
Relationship between density and surface temperature in heat insulation plates produced by pressurization of the raw materials mixed by compositions of Sample 1 at 2 atm ~ 30 atm is displayed in Fig. 4. As shown in the figure, surface temperatures were proportionately increased by an increase in heat transfer due to an increase in contact points as the specimen density was increased. The increase in surface temperatures in correspondence to a proportionate reduction in moving distance of heat with an increase in density of the heat insulation material was confirmed, in agreement with the previous report.15)
3.3. SiC particle size vs. surface temperature
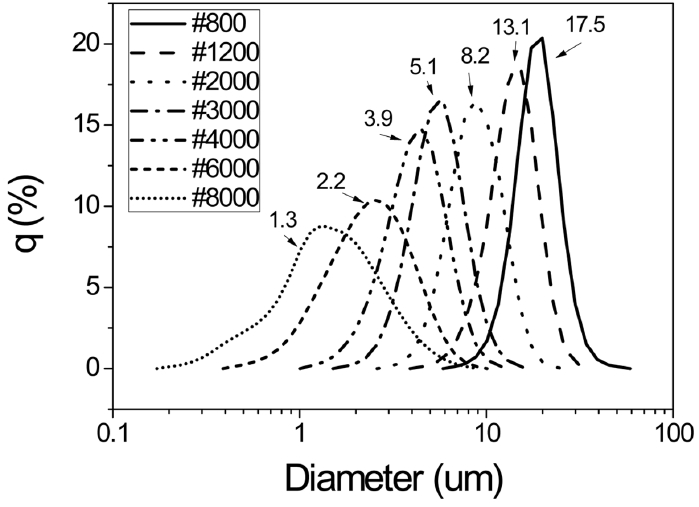
To investigate the effects of the particle size of SiC contained in heat insulation plates, surface temperatures of the plates were measured for 7 different sizes of SiC powders. Fig. 5 represents particle size distribution graphs for the SiC powders used, with an average particle diameter shown to be 1.3 ~ 17.5 μm.
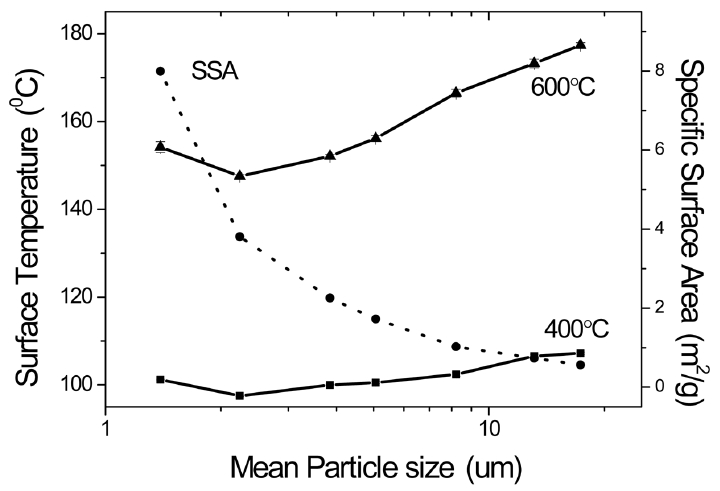
Heat insulation plates were prepared by using composition of Sample 1 (SiC 12.67%, glass fiber 4.67%, fumed silica 82.67%). Fig. 6 shows a change in the specific surface areas and the surface temperatures of plates as a function of the particle size of SiC powders. Specific surface areas were increased with a decrease in the particle size of SiC powders as expected. As the particle size of SiC was reduced down to 2.1 μm, the surface temperatures of heat insulation plates were reduced along with an increase in the specific surface area of SiC. While the surface temperature was expected to be the lowest in the specimen of 1.3 μm having the largest specific surface area, it was rather increased slightly. This is attributed to the effect of the particle size on Rosseland average extinction coefficients. The particle size is considered to be too small to sufficiently scatter the infrared spectrum.
Although the differences in surface temperatures as a function of SiC particle size were larger at the measurement temperature of 600°C rather than that of 400°C, the trend was similar. According to the report by Zhao et al., the Rosseland average extinction coefficient was large in the order of 3 μm>4 μm>5 μm>6 μm>1μm in average particle size at temperatures in the range of 400 ~ 600°C for SiC particles of fv = 3%. The Rosseland average extinction coefficient was drastically increased with an increase in temperatures for the size of 2 μm.5) According to the measurement results at 600°C, the Rosseland average extinction coefficient was large in the order of 3 μm > 2 μm ≒ 4 μm > 5 μm > 6 μm > 1 μm in average particle size. Thus, in the present study, the lowest surface temperature was observed in the heat insulation plate using a SiC particle size of 2.2 μm, although the surface temperature was expected to be lower in the plate using the particle size of 3.9 μm than 2.2 μm. The above results are slightly different from the results of Zhao et al., which is presumed to be a change in the extinction coefficient due to the shift of infrared radiation spectrum distribution as a function of measurement temperature by the differences in content, particle size and distribution of opacifiers. The previous study by Lee et al.,3) showed an increase in thermal conductivity with an increase in particle size by evaluating thermal conductivities with SiC particle size (10, 22, 31 μm) for the mixture of fumed silica powder by 75%, ceramic fiber by 10%, and SiC particles by 15%. Similar results were derived here although the composition in the present study was different. It appears that an increase in radiative heat transfer resulted as the particle size of SiC was increased to be larger than 2.2 μm which is considered combined effects of the peak intensity wavelength of blackbody radiation and the surface area of powders.
3.4. Inorganic binders vs. surface temperature and strength
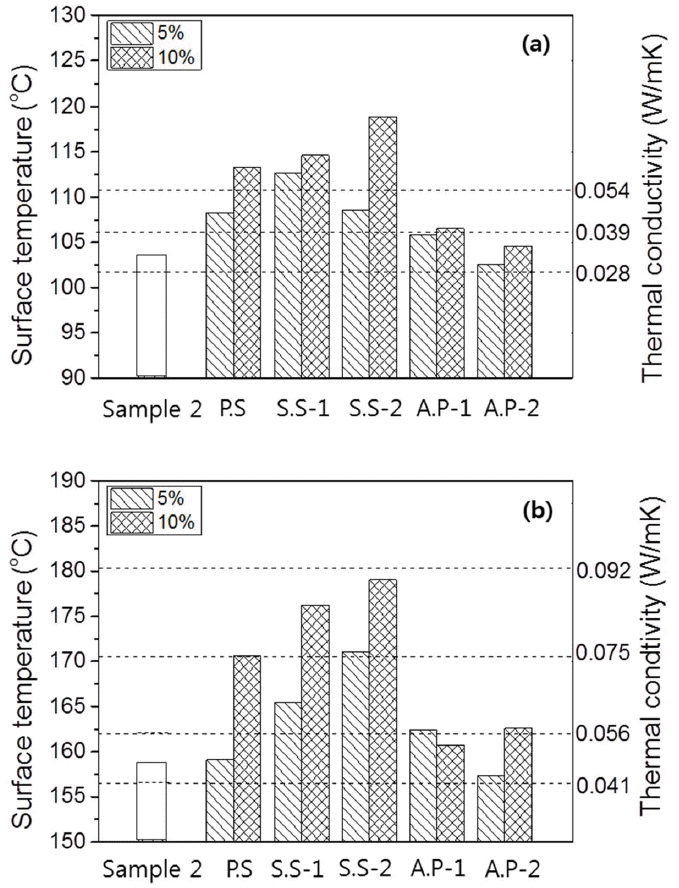
For the mechanical strength glass fiber reinforcement may be used but the amount should be limited to avoid the substantial increase in the thermal conductivity. For the high temperature heat insulation materials inorganic binders such as alkali silicates, phosphates, calcite salts etc. can be used instead of the organic binders for low temperature application. Selection of binders which enhance the mechanical strength but not deteriorate the thermal insulation is very important. Square plates were prepared for investigation of surface temperatures by adding 5% and 10% of each binder to the mixed powder having composition of Sample 2, composed of fumed silica by 80%, glass fiber by 10%, and SiC by 10%. Fig. 7 shows the measurement results for the surface temperature as a function of binder type by selecting the standard specimen without addition of binders (Sample 2 composition) and the specimens with 5 types of binders as shown in Table 1. The surface temperatures for 2 types of plates based on phosphates binder were lower than those for 3 types based on alkali silicates, among which the surface temperature was found to be the lowest exhibiting favorable heat insulation characteristics when A.P-2 binder was used. Thermal conductivities are expected to be increased when the gap between particles or between particle and fiber is connected by binder to have a structure for increased heat transfer. On the other hand, if the bonding structure is not dense, then heat transfer is considered to be suppressed so as to maintain low thermal conductivity values. Consequently, a loose bonding structure is predicted when the A.P-2 binder is used.
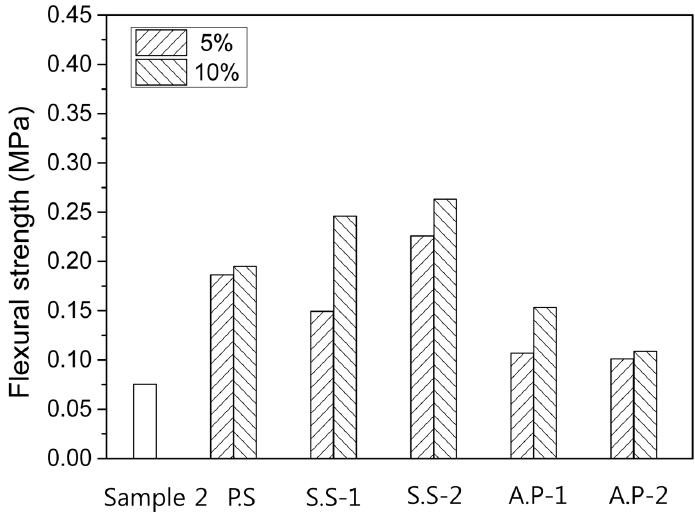
Figure 8 shows average values of flexural strengths measured for 6 times for the plates with inorganic binder type. As compared with the standard specimen without addition (Sample 2 composition), flexural strengths of the plates with addition of inorganic binders were increased, and an increase in the flexural strengths with an increase in the added amounts was observed for all inorganic binder types. The increase in flexural strengths were relatively larger when 3 types of binders based on alkali silicate were added in comparison with the case where phosphate binders were used, consistent with the surface temperature measurements shown in Fig. 7. The results confirmed that binders increase the surface temperatures of the specimens by increasing heat transfer due to an increase in contact points and at the same time contribute to an increase in flexural strengths by reinforcing bonding between particles, and between particle and fiber.
4. Conclusions
With fumed silica powder, glass fiber and SiC powder as the raw materials, green compacts of 300 × 300 mm plate having 10 different compositions were prepared and thermal conductivities were measured. Among those, five types of mixing ratios were selected to prepare 100 × 100 mm plates and surface temperatures were measured to confirm the correlation between thermal conductivity and surface temperature. Correlation coefficients between thermal conductivity and surface temperature at 400°C and 600°C were high as 0.96 and 0.97, respectively, suggesting that evaluation of heat insulation property was possible for the compacts of various mixing ratios by measuring the surface temperatures for green compacts of much smaller size than the specimens for evaluation of thermal conductivity by guarded hot plate method. The surface temperatures of the heat insulating plates decreased as the particle size of SiC opacifiers reduced from 17.5 μm to 2.1 μm and then slightly increased for 1.3 μm. The trends were similar although the differences in surface temperature as a function of SiC particle size were larger at the measurement temperature of 600°C than those at 400°C. The Surface temperature was low to exhibit good heat insulation properties when phosphate binder types were used compared to silicate types. The flexural strength was however correspondingly poorer.