Lee: Bloating Mechanism of Lightweight Aggregate with the Size
Abstract
The purpose of this study was to investigate the bloating mechanism of artificial lightweight aggregates with different sizes (ESA, effective surface area). Aggregates were produced using hard clay, stone sludge, and a bloating agent in order to observe the effect of the gradation of the artificial lightweight aggregates. Kerosene and amorphous carbon were used as bloating agent. The particle size of the produced aggregate ranged from 3 mm to 9 mm. With regard to the amount of bloating agent to be used, 2 ~ 6 parts by weight were used. The specific gravity, absorption rate, and the type of aggregates produced by rapid sintering at 1075 ~ 1200°C were determined. Microstructures were observed. When ESA had a value of 1 or below, kerosene, which has a high burning rate, was found to be advantageous for use as a bloating agent. When ESA had a value of 1 or above, carbon, which has a relatively low burning rate was found to be an advantageous bloating agent. It is thought that kerosene is more advantageous, as ESA decreases, for the production of aggregates having low water absorption rate.
Key words: Artificial lightweight aggregate, Bloating agent, ESA, Kerosen, Carbon
1. Introduction
Aggregates are generally classified into natural aggregate and artificial aggregate. Domestic natural aggregate resource is relatively abundant but the supply of aggregate has many potential problems for some reasons such as decrease in reserve that may be used as aggregate, extended distance from the sources, cost for recovering the pit site and rising logistics cost and thus dependency on alternative source such as marine resources tend to increase that results in quality problem with the aggregates. Artificial aggregate includes crushed stone aggregate, artificial lightweight aggregate and steel slag aggregate and the demand for artificial lightweight aggregate among them has been on the rise for high-story building or remodeling projects and a large market has been formed.
Artificial lightweight aggregate has been used since late 19th century in western countries and aggregates are produced mainly with natural material such as expanded shale or expanded clay, but not domestically because of lack of raw material. 1,2)
When producing artificial aggregates, kiln condition such as oxygen partial pressure or diffusion greatly influences on properties, the formation of black core inside the aggregate or reducing the weight. Oxygen diffusion which is one of the critical factor in reducing the weight is dependent on reaction temperature and aggregate surface thickness varies depending on oxygen diffusion through aggregate surface which is dependent on reaction time 3-6) and thus the more the oxygen that responds to aggregate the less the black core area. 7,8) Chemical formula when oxygen contacts organic matter is expressed as O 2(g) + 2[C+I](s)→I(s) + 2CO(g) Where, I refers to inorganic matter which forms the black core with carbon 3,5,6,9) and CO gas is discharged outside, but when aggregate surface is closed by sintering, gas is stick inside. Then CO(g) forms reduction atmosphere and reacts internally as Fe 2O 3 → FeO + 1/2O 2, thereby generating a closed pores and reducing the aggregate in weight. 10,11) Chen-Chiu Tsai 12) observed the change to physical properties of artificial lightweight aggregate due to the chemical composition of that during surface preparation and asserted that liquid viscosity of the surface is important for expanding the aggregate and V. Adell 13) asserted rapid sintering is more effective than slow sintering when expanding the aggregate. This means, reaction rate, reaction time and liquid on aggregate surface are important factor in reducing the weight of aggregate and correlations among them are considered the determinant in forming the black core and reducing the weight. However, particle-size of aggregate is also important factor besides the kiln condition and heating rate in producing lightweight fine aggregate, and the smaller the particle size of aggregate the larger the surface area and thus the reaction area of aggregate with oxygen becomes larger that would possibly lead to different characteristic from coarse aggregate.
This study thus is intended to identify the difference in expanding between fine aggregate and coarse aggregate of artificial lightweight under ordinary oxidation condition and rapid sintering test of artificial lightweight aggregate by particle size was conducted using amorphous carbon and kerosine so as to select the optimal bloating agent depending on aggregate size.
2. Experimental Procedure
2.1 Material
In this study, aggregate was produced primarily with red clay and stone sludge. Red clay was the clay used for producing red brick in market by Company C in Choongnam Province and stone sludge was the by-product from producing crushed sand, which accounts for about 20 ~ 30% in dry weight when producing crushed stone and annual production totals 3 million ton and main components are Si and Al with crystal phase similar with feldspars. Table 1 shows the components of the material used for the test. Bloating agent used for producing arttificial lightweight aggregate includes amorphous carbon (Sigma adrich, Korea) and kerosene.
2.2 ESA
ESA(Effective Surface Area) was introduced in a concept to express the relative surface area by particle size of aggregate.
It’s defined as ESI = S/ V. Where, S = Surface area of aggregate and V = Volume of aggregate. The shape of aggregate was assumed to be a complete globular shape and the equation is summarized as
ESI=3r, where, r is radius of aggregate. Table 2 shows the relationship between diameter of aggregate and ESI.
2.3 Preparation of material and test procedure
The material for the test was dried at 100°C for 24 h before crushing by pin mill to 180 μm or smaller and to identify the effect of particle size of aggregate and type of bloating agent on expanding, red clay, stone sludge and bloating agent were mixed at the ratio of 60 : 40 : 2, 60 : 40 : 4 and 60 : 40 : 6 with particle size of aggregate of 3, 4, 5, 6, 7 and 9 mm, respectively, before drying at 100°C for 24 h and putting in tube furnace at 1075 ~ 1200°C and maintaining for 15 minutes and then rapid sintering and quenching in the air before measuring the density and absorption rate (KSF 2503) and monitoring microstructure of pellet using Cam Scope (Sometech DCS 105, Korea)
3. Result and Discussion
3.1 Kerosene bloating agent
Figure 1 shows the density of artificial lightweight aggregate which was expanded using kerosene by rapid sintering temperature. No significant change in expansion characteristics was monitored despite of increase in bloating agent, but the more the bloating agent the less the density of artificial aggregate, which was attributable to difference in partial pressure between PCO and PCO2 depending on PO2 as revealed by Kang et al. 14) When PO2 is 10 −12.145 atm at 1400 k, PCO and PCO2 have same value and when PO2 becomes lower, CO becomes stable phase which is favorable in expanding process and thus, the more kerosene is added the lower PO2 and the greater PCO, resulting in more generation of gas that accelerates the expanding process.
Viewing expanding behavior of artificial lightweight aggregate by particle size in Fig. 1, rapid sintering of 3, 4 mm aggregate kept continuing till 1175°C, irrespective of the amount of kerosene and the density was slightly reduced at 1200°C and a very little expanding process was made. On the contrary, for rapid sintering of 7, 9 mm aggregate, the more the kerosene the lower the temperature that starts expanding process and more expanding process, resulting in more reduction in density. When it comes to 5,6 mm aggregate, it followed same sintering behavior as that of 3,4 mm till 1150°C and then bloating process started at 1150°C or higher and the density was reduced. Above result indicated that the smaller the aggregate size the more the difficulty with expanding process and according to C. M. Riley’s bloating mechanism of aggregate, 10) aggregate bloating is defined as expanding gas on closed surface of aggregate that generates closed pores internally. Stone sludge used for the test contains a considerable feldspar mineral which forms the liquid internally and accelerates densification of material that will close the surface of material. Then kerosene added as bloating agent starts burning and forming CO condition that converts Fe 2O 3 to Fe 3O 4 or FeO, making it black which is called a black core. A closed pores are expanded by such expanding gas as O 2 that reduces the density of aggregate. Thus it’s important to form the closed surface to capture the expanding gas and given that the smaller the aggregate size, the higher the temperature of closed surface, resulting in variation of density by particle size as shown in Fig. 1.
3.2 Amorphous carbon bloating agent
Figure 2 shows the density of artificial lightweight aggregate expanded using amorphous carbon bloating agent. On the contrary to kerosene bloating agent, no expanding and sintering behavior of aggregate in any size was monitored despite of increase in bloating agent ( Fig. 2(c)), which seemed to be attributable to no complete oxidation of carbon on aggregate surface because of increase in carbon which acts as inclusion during surface sintering that slows down surface densification and thus no expanding process occurs because of no capturing of gas and moreover, as carbon inside the aggregate that acts as inclusion during sintering process, no sintering occurs. Artificial aggregate using amorphous carbon as bloating agent ( Fig. 2(a)) starts expanding at higher temperature than kerosene ( Fig. 1(a)) with aggregates in large particle size, but density is reduced within temperature range with up to 4mm aggregate or less, which is attributable to relatively lower ignition point of kerosene than amorphous carbon. In case of aggregate in large particle size, surface area is smaller than volume and the material is closed before kerosene is completely burned and the smaller the diameter of aggregate the larger the surface area than volume and thus kerosene is burned before the surface of material is closed because of rapid heat conduction.
Thus, it would be difficult to monitor the reduction in density of aggregate when using kerosene as bloating agent for 3 ~ 4 mm aggregate and as shown with amorphous carbon, the smaller the particle size of aggregate the higher the temperature that will reduce the density but as combustion rate of amorphous carbon is relatively slow, reduction in density was monitored with 3 ~ 4 mm aggregate.
3.3 ESA
Viewing the result in Fig. 1 and Fig. 2, expanding temperature as well as expanding rate were dependent on size of artificial lightweight aggregate which indicates closed surface is essential for expanding process and is dependent by the size of aggregate, that is, surface area depending on volume of aggregate. Thus surface area per volume is defined as effective surface area (ESA) and Fig. 3 shows ESA and expanding temperature of artificial lightweight aggregate. When ESA is 1 o more, carbon is more favorable in bloating while kerosene is more favorable when kerosene is less than 1. The size of aggregates in globular shape when ESA is 1 is 6 mm and fine aggregate according to KS shall be 5mm or less and thus carbon is more favorable in bloating the fine aggregate of artificial lightweight aggregate.
3.4 Absorption rate
Figure 4 shows absorption rate by burning temperature of artificial lightweight aggregate which was expanded by adding bloating agent 2 parts by weight. Absorption rate of artificial lightweight aggregate using kerosene was less than that using carbon and the larger the aggregate size or the less ESA the greater the difference, which indicates that because the combustion rate of kerosene is faster, the less ESA the faster the evaporation, making closed surface of aggregate thicker, which is proven in Fig. 5 showing the section of artificial lightweight aggregate. Thus to produce the aggregate with less absorption rate, kerosene is more appropriate and reducing the content of carbon is important.
Black core was monitored on 3 ~ 4 mm aggregate among those using amorphous carbon as bloating agent as shown in Fig. 5 and density was reduced by expanded closed pores by black core while no black core was formed in 3 ~ 4 mm aggregate using kerosene as bloating agent, indicating no expanding process occurred.
4. Conclusions
As a result of monitoring expanding behavior of artificial lightweight aggregate depending on size of aggregate and type of bloating agent, the conclusion made is as follows.
The greater the kerosene added the less the density at same temperature which was attributable to increase in bloating agent burned that reduces partial pressure of oxygen and expansion of closed pores by gas which helps reduce the density. When it comes to aggregate using amorphous carbon, the more the bloating agent added the less the expanding and sintering because of carbon that acts as inclusion to hinder the movement When ESA is 1 or less, kerosene with faster combustion rate is more favorable as bloating agent while the carbon is more favorable when ESA is more than 1 To produce the aggregate with less absorption rate, kerosene is more favorable when ESA is less and reducing the content of carbon is important.
Acknowledgments
This work was supported by Kyonggi University Research Grant 2013.
Fig. 1
Specific gravity of aggregates bloated with kerosene: (a) 2 parts by weight, (b) 4 parts by weight, (c) 6 parts by weight. 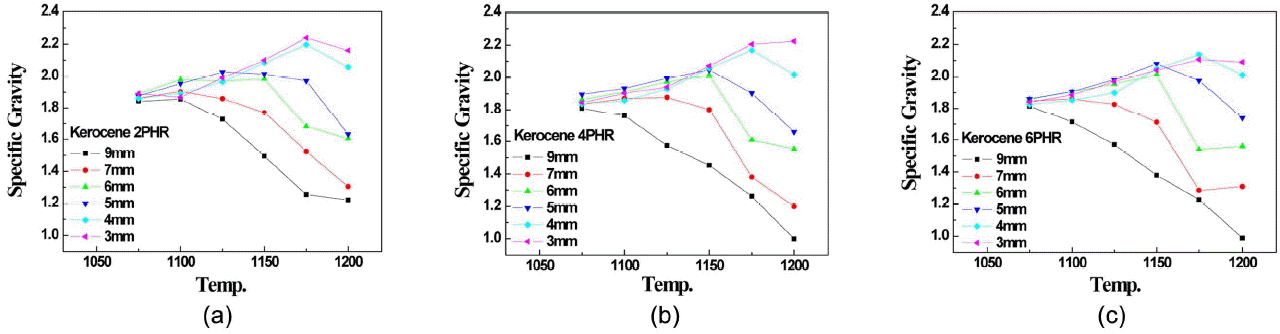
Fig. 2
Specific gravity of aggregates bloated with carbon: (a) 2parts by weight, (b) 4parts by weight, (c) 6parts by weight. 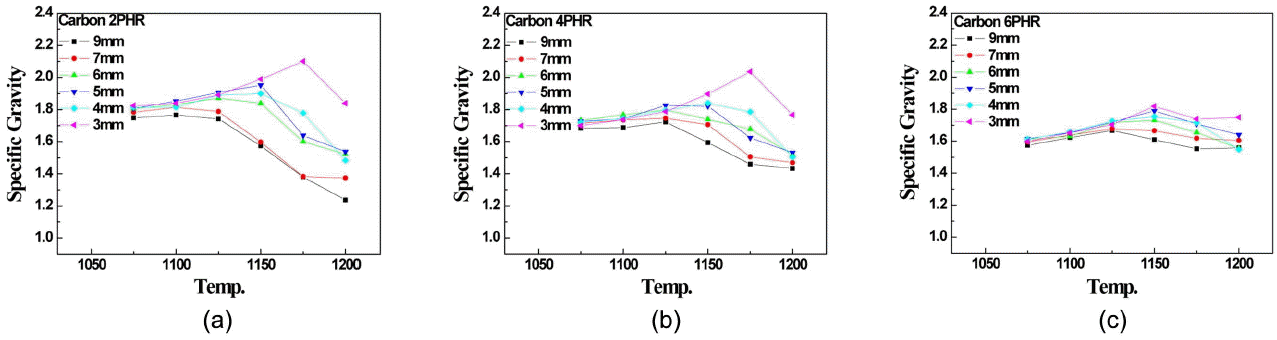
Fig. 3
Bloating temperatures as a function of ESA. 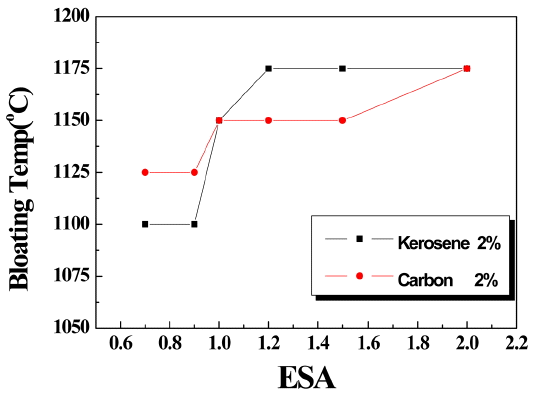
Fig. 4
Water absorption rate of various aggregates as a function of temperature. (a) 5,6 mm, (b) 7,9 mm. 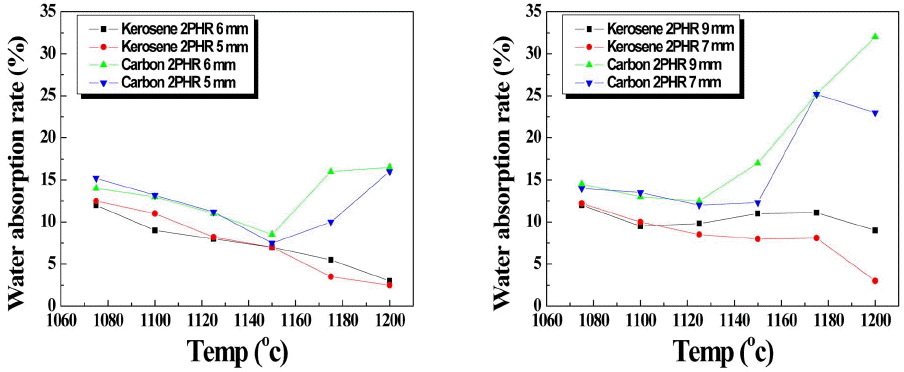
Fig. 5
Cross sectional images of various aggregates sintered at 1200°C. 
Table 1
Composition of Raw Materials (wt%)
|
Clay |
Stone Sludge(SS) |
|
SiO2
|
64.8 |
66.7 |
|
Al2O3
|
17.7 |
14.9 |
|
Fe2O3
|
7.2 |
2.3 |
|
CaO |
0.2 |
2.0 |
|
MgO |
0.7 |
1.1 |
|
Na2O |
0.2 |
3.8 |
|
K2O |
1.8 |
5.2 |
|
TiO2
|
1.0 |
0.3 |
|
P2O5
|
0.1 |
0.1 |
|
Ig-Loss |
6.3 |
3.6 |
Table 2
Relation between ESA and Size of Aggregates
|
ESA |
2 |
1.5 |
1.2 |
1 |
0.86 |
0.66 |
|
Size (mm) |
3 |
4 |
5 |
6 |
7 |
9 |
REFERENCES
1. YY. Choi, CH. Nam, BG. Kim, and BG. Kim, “Kiln for Production of Light Weight Aggregate from Coal Fly Ash,” J Korean Inst Resour Recycl, 16 [4] 61-7 (2007).
2. YJ. Kwon, and KG. Lee, “Analysis of Pore Characteristics on the Porous Body-Porosity Index of Ic and Is in Light Weight Aggregate,” J Korean Ceram Soc, 41 [2] 176-81 (2004).  3. A. Escardino, A. Barba, A. Blasco, and F. Negre, “Oxidation of Black Core during Firing of Ceramic Ware: 4 Relationship between Effective Diffusity of Oxygen through Oxidised Layer and Properties Characterising its Porous Structure,” Br Ceram Trans, 94 [3] 103(1995).
5. F. Negre, A. Barba, JL. Amoros, and A. Escarkino, “Oxidation of Black Core during the Firing of Ceramic Ware. II: Process Kinetics,” Br Ceram Trans, 91 [1] 5-11 (1992).
6. A. Barba, F. Negre, MJ. Orts, and A. Escardino, “Oxidation of Black Core during the Firing of Ceramic Ware. III. Influence of the Thickness of the Piece and the Composition of the Black Core,” Br Ceram Trans, 91 [2] 36-40 (1992).
7. YJ. Joo, and MS. Oh, “Unburned Carbon Combustion in the Production of Light Weight Sintered Fly Ash,”; pp. 91-96 in Proceeding of KSEE Conference, 2002.
8. SS. Cava, JA. Cerri, CA. Paskocimas, E. Longo, JA. Varela, T. Mendoca, and CG. Herter, “Supressing Black-Coring in Single-Fire Ceramic Tiles by Controlling Kiln Atmosphere,”; pp. 15-8 in Qualicer 98 : v World Congress on Ceramic Tile Quality, 1998.
9. A. Karamanov, P. Pisciella, C. Cantalini, and M. Pelino, “Influence of Fe3+/Fe2+ Ratio on the Crystallization of Iron-Rich Classes made with Industrial Wastes,” J Am Ceram Soc, 83 [12] 3153-57 (2000).  10. CM. Riley, “Relation of Chemical Properties to the Bloating of Clay,” J Am Ceram Soc, 34 [4] 121-28 (1951).  11. JY. Park, and YT. Kim, “Microstructural Observation of Artificial Aggregates at Various Sintering Atmospheres,” J Korean Cryst Growth Cryst Technol, 16 [2] 71-5 (2006).
12. C-C. Tsai, K-S. Wang, and I-J. Chiou, “Effect of SiO2-Al2O3-Flux Ratio Change on the Bloating Characteristics of Lightweight Aggregate Material Produced from Recycled Sewage Sludge,” J Hazard Mater, 134 [1] 87-93 (2006).  13. V. Adell, CR. Cheeseman, A. Doel, A. Beattie, and AR. Boccaccini, “Comparison of Rapid and Slow Sintered Pulverised Fuel Ash,” Fuel, 87 [2] 187-95 (2008).  14. SH. Kang, KG. Lee, YT. Kim, and SG. Kang, “Effect of Chemicophysical Properties of Carbon on Bloating Characteristics of Artificial Lightweight Aggregates Using Coal Ash,” Ceram Trans, 232 35-42 (2012). 
|
|
















